Introduction
Human cytomegalovirus (HCMV) is a member of the Herpesviridae family and beta Herpesvirinae subfamily. Also, HCMV infection can induce latency in white blood cells for life long. This persistent infection involved 50-90% of adults in the United States [1]. This virus can trigger a great range of symptoms in children and the elderly but is mostly asymptomatic in immunocompetent adults [2, 3]. Studies in the United States indicated that 30-40% of children aged 6-11 years are seropositive for HCMV [4]. This virus is distributed worldwide, but the prevalence varies from 45% in developed countries to 100% in developing countries. Also, HCMV infection mostly occurs in children less than five years in developing countries [2]. HCMV is transmitted through oral and sexual routes, blood transfusion, and organ transplantation, and also, the virus could be isolated from sexual secretions, urine, sperm, and amniotic fluid [5].
Also, sexual transmission is the major reason for increasing seropositive adult patients. The rate of seropositivity for HCMV in adults was increased in the cases with a history of other sexually transmitted diseases (STDs). It has been indicated that HCMV can induce mutations, evade the immune system, and manipulate the cell cycle [6, 7]. In reproductive medicine, STDs cause major problems, and some of them could be mortal or incurable [8-10]. Furthermore, there are different reasons for infertility in men, but 50% of men's infertility problems are idiopathic [11-16]. Therefore, it has been suggested that these infertility cases in men could be due to asymptomatic infections [8, 12, 13, 17, 18]. STDs have been introduced as a cause for idiopathic infertilities. Viral infections can play essential roles in infertility directly by affecting spermatogenesis and indirectly by altering the immune system [11, 16, 17]. Viral infections can affect the sperm's parameters, such as motility or cunt. Furthermore, the quality of sperms was affected in asymptomatic people without infertility [16, 19]. Regarding the critical role of viruses in the induction of infertility and the importance of herpesviruses in this field, the current study aimed to assess the prevalence of HCMV in sperm samples of infertile men.
Materials and Methods
Patient selection and sample preparation
Patients enrolled in this cross-sectional study were asymptomatic infertile men referred to the infertility centers affiliated to Iran University of Medical Sciences between 2017 and 2018. This study was approved by the Ethical Committee of Iran University of Medical Sciences (IR.IUMS.REC.1396.30512). Moreover, written consent was obtained from enrolled patients. Semen specimens were collected in tubes and stored in -20ºC until further analysis. According to the manufacturer, the DNA fragmentation index (DFI) was performed by the Sperm DNA Fragmentation Assay Kit (SDFA, ACECR, Academic center for education, culture, and research, Tehran, Iran)'s protocols. Other parameters of sperm analysis include sperm motility, count, and morphology, were determined based on the World Health Organization criteria [20].
DNA extraction
According to the manufacturer's protocol, DNA was extracted from samples using Tissue Genomic DNA Extraction (Favorgene, Taiwan) kit. The quantity of extracted DNA was evaluated by the NanoDrop ND-1000® spectrophotometry (Thermo Fisher Scientific Inc., USA). Also, for approving the human spermatozoid genome's isolation procedures in samples, a conventional polymerase chain reaction (PCR) assay was performed for the beta-globin gene.
PCR for beta-globin gene
A conventional PCR assay was performed for the beta-globin gene in the Bio-Rad thermocycler (T100™ Thermal Cycler) with the following thermal conditions: 10 min at 95˚C as initial denaturation, then, 35 cycles of 1 min at 94˚C, the 30 s at 55˚C, 40 s at 72˚C, and one final extension step at 72˚C for 10 min. For PCR product visualization, gel electrophoresis in 1.5% agarose and ethidium bromide staining were used. The reaction mixture was as follows: 12.5 µl 2X Amplicon Master Mix (Amplicon, Denmark), 25-50 ng of template DNA, 1 µl of each primer (10 pmol/µl), and sterilized deionized water added to round out to 25 µl for each test.
Detection of HCMV
The presence of HCMV DNA was evaluated using real-time PCR. The real-time PCR was performed using specific primers and probes to detect the HCMV- UL55 gene (Table 1). The reaction mix contained 25-50 ng of template DNA, 10 pmol/µl each primer, and 8 µl 2X master mix (qPCR for Prob, Yekta Tajhiz Azma, Iran) and distilled water added to 16 µl final volume. The real-time PCR was performed in Rotor-Gene-Q 6000 thermocycler (Corbett, Australia) by 45 cycles includes 30 s in 94˚C and 30 s in 60˚C.
Statistical analysis
Statistical analysis was performed by SPSS version 22 (SPSS Inc., Chicago, IL, USA), and Fisher's exact test and chi-square test were used to assess the results. The p-values less than 0.05 were considered statistically significant in this study.
Results
The mean age of 82 males was 37.3 ± 6.1 years. Assessment of sperm parameters showed that the mean sperm count was 21.5 ± 13 sperm in each milliliter. Also, the mean sperm motility was 33.65±20.14%. The morphology assessment of the sperms showed normal morphology in 49.2% of the patients. Furthermore, the mean DFI was 27.2 ± 1.2, and the DFI was poor in 26.8% of the enrolled patients (Table 2). The result of PCR for the beta-globin gene showed efficient isolation of genomic DNA from semen samples (Figure 1). The real-time PCR assay showed HCMV infection in 8 (11%) patients (Fig. 2). The results of the real-time PCR assay are summarized in Table 3. The study results for HCMV and different sperm parameters could not show significant association except in sperm motility (p = 0.008).
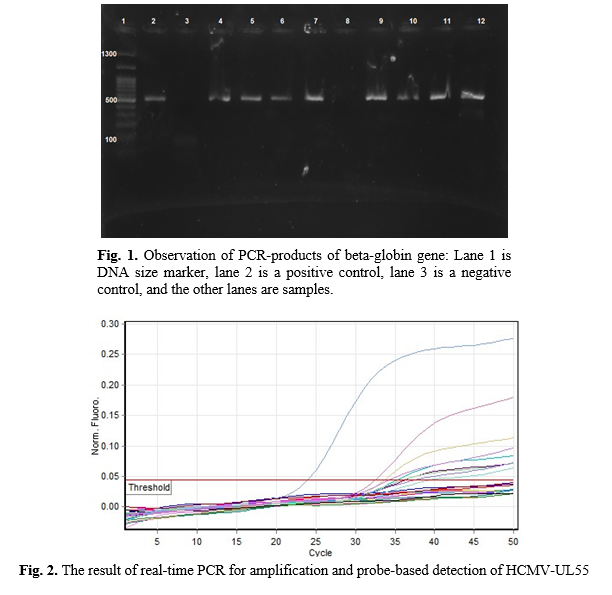
Table 1. The features of the primers and the probe used in this study
| Target |
sequence |
Product size |
| Beta-globin gene |
Forward |
3' TCAACCCTACAGTCACCCAT 5' |
501 bp |
| Reverse |
3' CTAACAATTACGAACAGCAATGAG 5' |
| HCMV-UL55 |
Forward |
3' TGGGCGAGGACAACGAA 5' |
66 bp |
| Reverse |
3' TGAGGCTGGGAAGCTGACAT 5' |
| Probe |
3'- 6- FAM- TCGGCAACCACCGCACTG 5' |
Table 2. The DNA fragmentation index results
| Frequency |
Number |
Chance of conception |
Sperm chromatin dispersion |
| 18.4 |
15 |
Excellent |
DFI < 15 |
| 39 |
32 |
Good |
15-25 |
| 15.8 |
13 |
Fair |
26-30 |
| 26.8 |
22 |
Poor |
DFI > 30 |
DFI= Fragmentation index
Table 3. HCMV real-time PCR and its association with other sperm parameters
| Motility (%) |
Count |
DFI |
Mean age |
CMV |
| 50.1 ± 1.9 |
27.8 ± 10.5 |
22.1 ± 1 |
38.7 ± 5.4 |
CMV positive |
| 31.5 ± 1.9 |
20.7 ±13.1 |
27.8 ± 1.6 |
37.1 ± 6 |
CMV negative |
| 0.008* |
0.124 |
0.291 |
0.446 |
P- value |
Discussion
It has been indicated that 15-25% of couples have faced infertility problems, and 20-50% of all these infertility problems are related to males [11, 18]. There are different reasons for infertility, and the most prevalent condition (35% of all cases) in males is due to varicocele. Furthermore, endocrine diseases, anti-sperm antibodies, chemotherapy, radiotherapy, and STDs can affect males’ fertility. Also, 50% of male infertility conditions are idiopathic [11-14]. Investigations suggested that sexually transmitted pathogens can be preserved for long durations in the male reproductive system and its ducts [11, 17, 18]. Also, it has been suggested that infertility in males can be due to asymptomatic infections in the reproductive system [8, 10, 11, 18]. The role of different viruses in infertility has been investigated [19, 21, 22]. Furthermore, herpesviruses' role in infertility was assessed in various studies [11, 19, 23, 24].
Monavari et al. measured the prevalence of herpes simplex virus (HSV)-1 and HSV-2 in semen samples of asymptomatic infertile men using real-time PCR. According to their results, HSV-1 and HSV-2 DNA were found in 16 (22.9%) and 10 (14.3%) of 70 semen samples, respectively. They concluded that infection with HSV is not associated with sperm motility and morphological defects. Instead, it is related to lower sperm count in the seminal fluid [24].
In another study, Behboudi et al. did not find any association between the detection of Varicella-zoster virus, HCMV,
adeno-associated virus (AAV), and human papillomaviruses (HPV), DNA in semen samples and variations in semen parameters related to fertility [21]. Moghimi et al. conducted a case-control study to determine the correlation between impairment of sperm quality in infertile Iranian men and seminal high-risk HPV infection [25]. According to their results, high-risk HPV was detected in 11.4% of infertile males, and all fertile men were HPV-negative. They concluded that HPV infection might be a risk factor for male infertility.
Kapranos et al. investigated the role of viruses in infertility, and they reported that the prevalence of viral infection in infertile men is 56% [26]. Also, they reported a prevalence of 7% HCMV in specimens of infertile men. In
Kapranos's study, there was no statistically significant difference between mean sperm count and motility. Also, Chen et al. reported that viral infection in infertile men with ages ranged from 21-49 years was 39% [18], while the prevalence of HCMV in this study was 22%. However, in Chen's study, the sperm parameters and infection with viruses did not show a statistically significant difference. In the study conducted by
Naumenko et al., the prevalence of HCMV was reported as 5.2%, and they have investigated the effects of HCMV and Epstein-Barr virus in urogenital inflammation cases [17]. Furthermore, Bezold et al. have shown a significant association between sperm motility in men ranged from 22-55 years old and infection with sexually transmitted pathogens [19].
The current study results indicated that the mean age of the infertile men was 37.3 ± 6.1 years, and the real-time PCR assay showed HCMV infection in 8/82 (11%) of patients. Also, assessment of HCMV and different sperm parameters showed a significant association in sperm motility (p=0.008). The mentioned studies support the present study results, and the differences between the results of these studies and the present study can be justified by considering different methods and sample sizes used. Also, the effects of co-infections in this field and its association with sperm parameters can be significant [19]. Limitations of the current study include the sample size and the lack of assessing the other herpesviruses or more sperm parameters.
Conclusion
This study concludes that infection with HCMV can be important in sperm motility and the prevalence of HCMV in infertile men is 11%. We suggest further investigations in this field to have more precise results.
Conflict of Interest
The authors have no conflict of interest.
Acknowledgments
The study was financially supported by the Iran University of Medical Sciences (Grants No. 30512).
References
- Benson PJ, Smith CS. Cytomegalovirus prostatitis. Urology 1992; 40(2): 165-67.
- Plosa EJ, Esbenshade JC, Fuller MP, Weitkamp JH. Cytomegalovirus infection. Pediatr Rev. 2012; 33(4): 156-63.
- Almeida L, Azevedo R, Amaku M, Massad E. Cytomegalovirus seroepidemiology in an urban community of São Paulo, Brazil. Rev Saude Publica. 2001; 35(1): 124-29.
- Staras SA, Dollard SC, Radford KW, Flanders WD, Pass RF, Cannon MJ. Seroprevalence of cytomegalovirus infection in the United States, 1988-1994. Clin Infect Dis. 2006; 43(9): 1143-151.
- Tarabadi F, Babaeie G, Hashemi E, Boroumand B, Shaigan M. Compmison of Anti-CMV (IGG-IGM) serologic prevalence among controls and patients. Hakim Res J. 2002; 4(4): 243-47.
- Castillo JP, Kowalik TF. Human cytomegalo-virus immediate early proteins and cell growth control. Gene 2002; 290(1-2): 19-34.
- Doniger J, Muralidhar S, Rosenthal LJ. Human cytomegalovirus and human herpesvirus 6 genes that transform and transactivate. Clin Microbiol Rev. 1999; 12(3): 367-82.
- Wortley PM, Hammett TA, Fleming PL. Donor insemination and human immunodeficiency virus transmission. Obstet Gynecol. 1998; 91(4): 515-18.
- Moore DE, Ashley RL, Zarutskie PW, Coombs RW, Soules MR, Corey L. Transmission of genital herpes by donor insemination. JAMA 1989; 261(23): 3441-443.
- Berry WR, Gottesfeld RL, Alter HJ, Vierling JM. Transmission of hepatitis B virus by artificial insemination. JAMA 1987; 257(8): 1079-1081.
- Neofytou E, Sourvinos G, Asmarianaki M, Spandidos DA, Makrigiannakis A. Prevalence of human herpes virus types 1–7 in the semen of men attending an infertility clinic and correlation with semen parameters. Fertil Steril 2009; 91(6): 2487-494.
- Greenberg SH, Lipshultz LI, Wein AJ. Experience with 425 subfertile male patients. J Urol 1978; 119(4): 507-10.
- Skakkebaek NE, Giwercman A. Pathogenesis and management of male infertility. Lancet 1994; 343(8911): 1473-479.
- Dejucq-Rainsford N, Jegou B. Viruses in semen and male genital tissues-consequences for the reproductive system and therapeutic perspectives. Curr Pharm Des. 2004; 10(5): 557-75.
- World Health Organization. WHO laboratory manual for the examination of human semen and sperm-cervical mucus interaction: Cambridge university press; 1999.
- Garolla A, Pizzol D, Bertoldo A, Menegazzo M, Barzon L, Foresta C. Sperm viral infection and male infertility: focus on HBV, HCV, HIV, HPV, HSV, HCMV, and AAV. J Reprod Immunol. 2013; 100(1): 20-29.
- Naumenko V, Tyulenev Y, Kurilo L, Shileiko L, Sorokina T, Evdokimov V, et al. Detection and quantification of human herpes viruses types 4-6 in sperm samples of patients with fertility disorders and chronic inflammatory urogenital tract diseases. Andrology 2014; 2(5): 687-94.
- Chen M, Cai LY, Kanno N, Kato T, Lu J, Jin F, et al. Detection of human herpesviruses (HHVs) in semen of human male infertile patients. J Reprod Dev. 2013; 59(5): 457-62.
- Bezold G, Politch JA, Kiviat NB, Kuypers JM, Wolff H, Anderson DJ. Prevalence of sexually transmissible pathogens in semen from asymptomatic male infertility patients with and without leukocytospermia. Int J Fertil Steril. 2007; 87(5): 1087-1097.
- World Health Organization. WHO laboratory manual for the examination and processing of human semen, 5th ed. 2010.
- Behboudi E, Mokhtari-Azad T, Yavarian J, Ghavami N, Seyed Khorrami SM, Rezaei F, et al. Molecular detection of HHV1-5, AAV and HPV in semen specimens and their impact on male fertility. Hum Fertil. 2019; 22(2): 133-38.
- Nasseri S, Monavari SH, Keyvani H, Nikkhoo B, Roudsari RV, Khazeni M. The prevalence of human papilloma virus (HPV) infection in the oligospermic and azoospermic men. Med J Islam Repub Iran 2015; 29(1): 1001-1006.
- Salehi-vaziri M, Monavari S, Khalili M, Shamsi-Shahrabadi M, Keyvani H, Mollaei H, et al. Detection of HSV-1 DNA in the semen of infertile men and evaluation of its correlation with semen parameters in Iran. Iran J Virol. 2010; 4(2): 1-6.
- Monavari SH, Vaziri MS, Khalili M, Shamsi-Shahrabadi M, Keyvani H, Mollaei H, et al. Asymptomatic seminal infection of herpes simplex virus: impact on male infertility. J Biomed Res. 2013; 27(1): 56-61.
- Moghimi M, Zabihi-Mahmoodabadi S, Kheirkhah-Vakilabad A, Kargar Z. Significant correlation between high-risk HPV DNA in semen and impairment of sperm quality in infertile men. Int J Fertil Steril. 2019; 12(4): 306-309.
- Kapranos N, Petrakou E, Anastasiadou C, Kotronias D. Detection of herpes simplex virus, cytomegalovirus, and Epstein-Barr virus in the semen of men attending an infertility clinic. Fertil Steril. 2003; 79: 1566-570.









































 , Somayeh Sadat Moochani
, Somayeh Sadat Moochani 
 , Nazila Yamini
, Nazila Yamini 
 , Davod Javanmard
, Davod Javanmard 
 , Arezoo Marjani
, Arezoo Marjani 
 , Alireza Tabibzadeh
, Alireza Tabibzadeh 
 , Ahmad Tavakoli
, Ahmad Tavakoli 
 , Seyed Hamidreza Monavari *
, Seyed Hamidreza Monavari * 


