Tue, Apr 7, 2026
[Archive]
Volume 11, Issue 2 (May 2024)
IJML 2024, 11(2): 136-145 |
Back to browse issues page
Download citation:
BibTeX | RIS | EndNote | Medlars | ProCite | Reference Manager | RefWorks
Send citation to:



BibTeX | RIS | EndNote | Medlars | ProCite | Reference Manager | RefWorks
Send citation to:
Ahmarinezhad Z, Deyhim M R, Lesan Pezeshki M, Najafi M T, Khoshnaghsh F. Evaluation of Lipid Peroxidation, Nitric Oxide Metabolites, and Plasma Total Homocysteine Concentration in Patients with Chronic Kidney Disease. IJML 2024; 11 (2) :136-145
URL: http://ijml.ssu.ac.ir/article-1-528-en.html
URL: http://ijml.ssu.ac.ir/article-1-528-en.html
Zeynab Ahmarinezhad 
 , Mohammad Reza Deyhim *
, Mohammad Reza Deyhim * 
 , Mahbub Lesan Pezeshki
, Mahbub Lesan Pezeshki 
 , Mohammad Taghi Najafi
, Mohammad Taghi Najafi 
 , Fahimeh Khoshnaghsh
, Fahimeh Khoshnaghsh 


 , Mohammad Reza Deyhim *
, Mohammad Reza Deyhim * 
 , Mahbub Lesan Pezeshki
, Mahbub Lesan Pezeshki 
 , Mohammad Taghi Najafi
, Mohammad Taghi Najafi 
 , Fahimeh Khoshnaghsh
, Fahimeh Khoshnaghsh 

Iranian Blood Transfusion Research Center, High Institute for Research and Education in Transfusion Medicine, Tehran, Iran
Full-Text [PDF 240 kb]
(278 Downloads)
| Abstract (HTML) (781 Views)
Table 2. Evaluation of Blood Biochemical Parameters in CKD Patients Compared with the Control Group
Discussion
References
Full-Text: (316 Views)
Introduction
Chronic kidney disease (CKD), often referred to as chronic kidney failure, is characterized by a progressive decline in kidney function over time [1]. CKD results in numerous fatalities worldwide each year. Among CKD patients, cardiovascular disease is the primary cause of death, and its prevalence has been on the rise globally [2]. Established risk factors, including diabetes, hypertension, and dyslipidemia, have been shown to have a significant correlation with cardiovascular disease in patients with CKD.
One of the newly identified risk factors is oxidative stress, characterized by an excess of reactive oxygen species (ROS) and/or a diminished capacity for antioxidant defense. This phenomenon has been extensively documented and researched in patients with uremia.
It may play a role in the development of atherosclerosis, cardiovascular incidents, and various complications associated with CKD, including endothelial cell dysfunction, anemia, and protein-energy malnutrition [3].
Free radical reactions, particularly lipid peroxidation, are regarded as significant contributors to the development of various diseases, especially CKD [4, 5]. The progression of CKD may lead to an increase in lipid peroxidation and a reduction in antioxidant defenses, potentially resulting in oxidative damage to the kidneys of patients suffering from chronic renal failure [6]. One significant consequence of membrane lipid oxidation, primarily taking place in mitochondria, is the impairment of normal endothelial function, a critical element in cardiovascular diseases linked to CKD [7]. This phenomenon arises from the interaction of ROS with the double bonds present in unsaturated lipids, ultimately resulting in the generation and accumulation of lipid radicals. This alteration in membrane lipid structure leads to the formation of various products, including reactive electrophiles such as epoxides and aldehydes [4, 5, 8]. Malondialdehyde is a significant and primary byproduct of lipid peroxidation, serving as an important biomarker for evaluating the extent of lipid peroxidation [4, 5, 9].
Nitric oxide (NO) and its derivatives, nitrite and nitrate, serve as significant signaling molecules involved in numerous physiological and pathological conditions. Their primary functions can be categorized into three main areas:
the regulation of blood flow within the vascular endothelium (vascular homeostasis), neurotransmission, and mechanisms of host defense. The role of NO in the kidneys is multifaceted, influenced by its concentration, the specific site of release, and the duration of its action. Endothelial damage and the resulting dysfunction of the microcirculation are likely initial events in renal hypoxia, with disturbances in NO-related pathways playing crucial roles. Furthermore, NO metabolites, such as nitrite and nitrate, may serve as potential biomarkers for evaluating vascular endothelial damage in patients with CKD [10].
In recent years, research has focused on the direct correlation between changes in homocysteine metabolism and the unfavorable outcomes associated with kidney diseases. Homocysteine is a sulfur-containing non-proteinogenic amino acid that results from the demethylation of the essential amino acid methionine (Met). It serves as an intermediate in the biosynthetic pathway that facilitates the conversion of Met into cysteine. The metabolism of homocysteine takes place through
two primary pathways: remethylation and transsulfuration [11, 12, 13]. Homocysteine is recognized for its transsulfuration process, which occurs in the kidneys. Consequently, notable changes in the enzymes and mechanisms associated with transsulfuration significantly elevate plasma homocysteine levels while concurrently causing impairment in renal and cardiovascular functions. A moderate rise in plasma homocysteine levels can be detected from the early stages of CKD, coinciding with deterioration in renal function. This change is regarded as a critical element in the advancement of kidney diseases, particularly CKD [14].
Consequently, it is crucial to identify and utilize biomarkers that can facilitate the early diagnosis of kidney damage. In this research, we assessed several of these markers in individuals diagnosed with CKD.
Materials and Methods
Sample collection and preparation
The present study is a case-control study. The sample size in this study was calculated based on the significance level of 0.05 and the power of 80%, the minimum required size of 60 samples in each group. Thus, blood samples were taken from 38 male and 23 female patients diagnosed with CKD, with an average age of 58.9 ± 15.9 and 62.13 ± 13.43, respectively. At the same time, 40 healthy male individuals and 22 healthy female individuals, with an average age of 60.33 ± 10.62 and 59.3±6.64, respectively, were concurrently selected as the control group. These individuals had no prior history of any diseases, including hypertension, diabetes, infections, or kidney disease. They also had no history of smoking. Also, neither the case group nor the control group took any antioxidant drugs or other drugs that could interfere with the research parameters. All patients were 4 grade of CKD and were treated in the nephrology department of the hospital. Informed consent was obtained from the patients and subjects involved in the current study. Ethical committee approval has also been obtained. To perform tests first, 10 ml of whole blood sample was taken from each person in each group and transferred to two tubes containing EDTA-anticoagulant and without anticoagulant. Then, blood samples taken from patients were centrifuged at 4000 rpm for 10 minutes to obtain the required plasma and serum for the experiments. Hematological parameters were performed on whole blood samples.
Measurement of hematological parameters
Erythrocyte count, hematocrit, hemoglobin concentration, and other erythrocyte indices such as mean corpuscular hemoglobin (MCH), mean corpuscular hemoglobin concentration (MCHC), and mean corpuscular volume (MCV) were measured by cell counting device (Sismex, K-1000, Japan).
Measurement of biochemical parameters
All measurements of biochemical parameters, including glucose, urea, calcium, and phosphorus concentrations, were performed on a serum sample by a Chemistry autoanalyzer (Hitachi 902, Japan). Thus, the Concentration of urea was measured by the urease method, creatinine by the Jaffe reaction, calcium by the Arsenazo method, and the concentration of phosphorus was measured by the phosphomolabate method according to the kit protocol. Sodium and potassium concentrations were also measured using a flame photometer (Eppendorf, Efox 5054, Germany).
Plasma Homocysteine assessment
A plasma sample was utilized to assess homocysteine levels. The concentration of homocysteine was determined following the protocol provided by the kit (Axis-Shield, UK). This procedure involves two distinct steps.
Step 1 (Reduction reaction):
At this stage, plasma protein-bound homocysteine is first reduced to free homocysteine by tris (2-carboxyethyl) phosphine.
Step 2 (Enzymatic conversion reaction):
In this step, homocysteine is converted to cystathionine by cystathionine beta-synthase, and then cystathionine is converted to Pyruvate by cystathionine beta-lyase. Following this, pyruvate is converted to lactate by a lactate dehydrogenase enzymatic reaction (NADH is used as a cofactor in this reaction). In this reaction, the optical density (OD) of NADH to NAD converter, which is proportional to the initial concentration of homocysteine, is read using an autoanalyzer (Cobas Mira, Roche, Germany).
Malondialdehyde assessment
Malondialdehyde is the primary byproduct resulting from the peroxidation of polyunsaturated fatty acids. The concentration of malondialdehyde is measured by the reaction of thiobarbituric acid [15]. In this reaction, after the proteins were precipitated by trichloroacetic acid, the unsaturated fatty acids were oxidized with thiobarbituric acid, resulting in a color complex whose adsorption rate at 532 nm was read. This adsorption rate is converted to concentration (nmol) using a standard curve.
Measurement of nitrite/nitrate metabolites
The content of NO metabolites was assessed using the classic Griess spectro-photometric assay [16]. Briefly, the nitrate content of the sample was reduced to nitrite using vanadium trichloride. Griess reagent was added, and the samples were incubated at 37 °C for 45 minutes. Optical density (OD) was assayed using an enzyme-linked immunosorbent assay reader (Hiperion, Germany) at 540 nm. The adsorption rate is converted to concentration (µmol) using a standard curve.
Statistical Analysis
SPSS v.22.0 software (SPSS Inc., Chicago, IL, USA) was used to analyze data. The Kolmogorov-Smirnov test assayed data normal distribution. Independent sample t-test and Mann-Whitney were performed to compare groups at each time point. The results were displayed as mean standard deviation (SD), and P < 0.05 was considered significant.
Results
Hematological parameters
As indicated in Table 1, the erythrocyte count, hemoglobin levels, and hematocrit values in patients with CKD were significantly lower when compared to the control group (p < 0.01). Additionally, the erythrocyte indices, which encompass MCV, MCH, and MCHC, exhibited significant alterations in CKD patients relative to the control group (p = 0.03, p = 0.02, p = 0.01), respectively.
Biochemical parameters
The findings displayed in Table 2 indicate that the concentrations of urea, creatinine, phosphorus, and potassium were significantly elevated in individuals with CKD when compared to the control group (p = 0.0001, p = 0.0001, p = 0.001, p = 0.01), respectively. Conversely, the concentrations of sodium and calcium were significantly reduced in the CKD group relative to the control (p < 0.01).
Homocysteine concentration
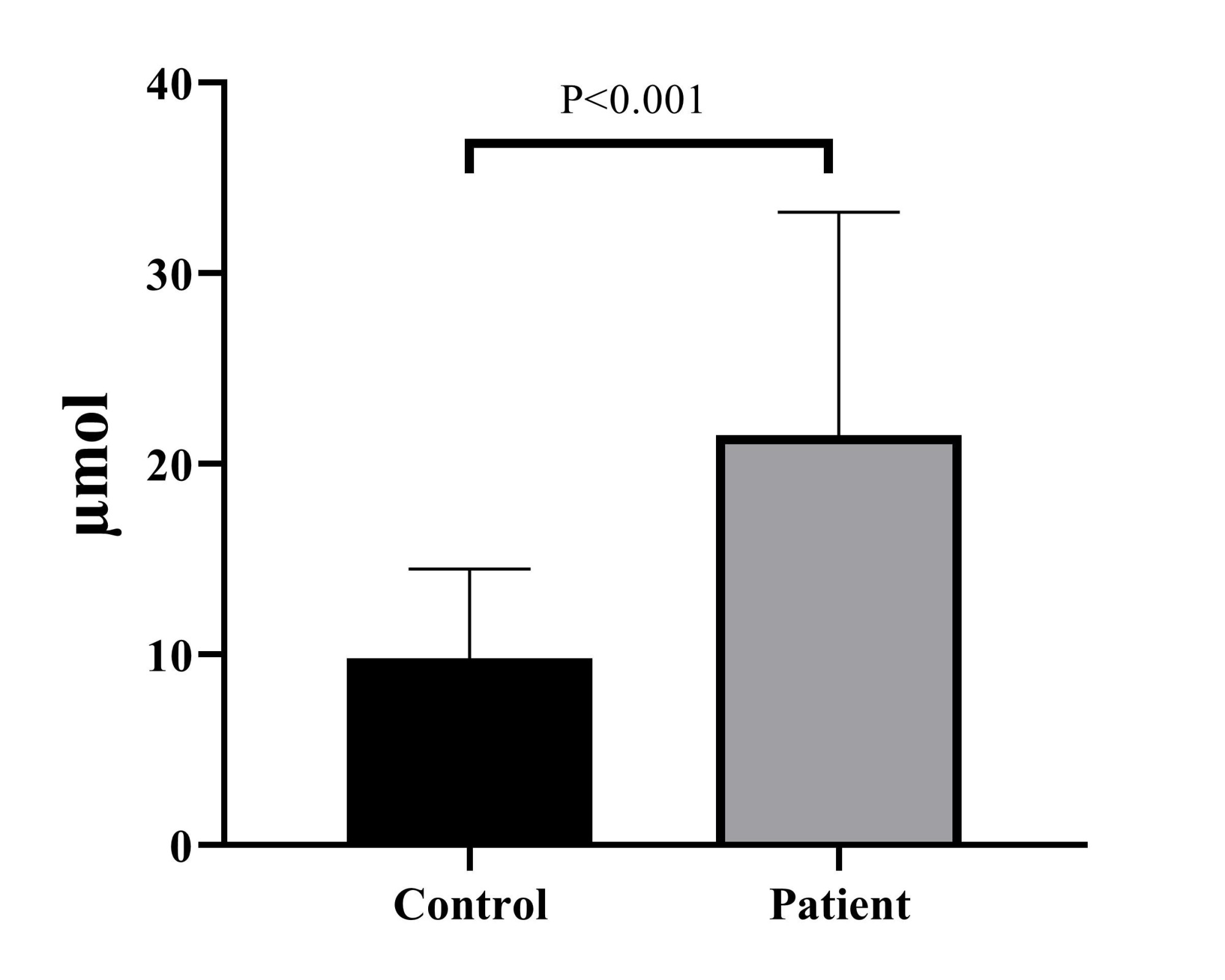
The findings from the enzymatic assessment of homocysteine indicated that the concentration of homocysteine was typically elevated in patients with CKD (21.52 ± 11.69) compared to the control group (9.8 ± 4.7), which was statistically significant (p < 0.001) (Fig. 1).
Malondialdehyde concentration
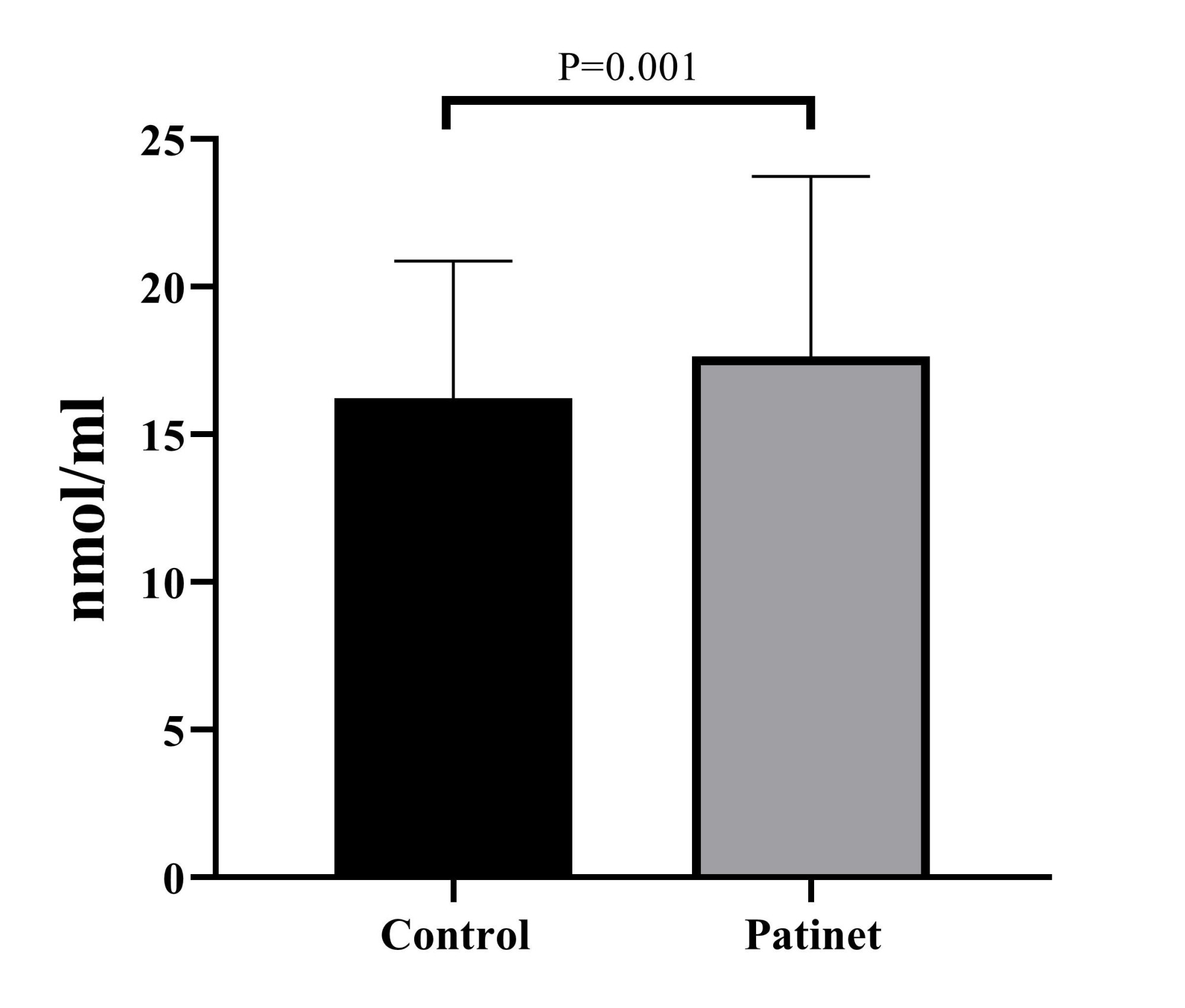
The results showed an increase in malondialdehyde concentration in the CKD group (17.64 ± 6.1) compared to the control group (16.22 ± 4.66), which, according to the statistical analysis of the t-test, this difference was found to be statistically significant (p = 0.01) (Fig. 2).
NO metabolites (nitrite/nitrate)
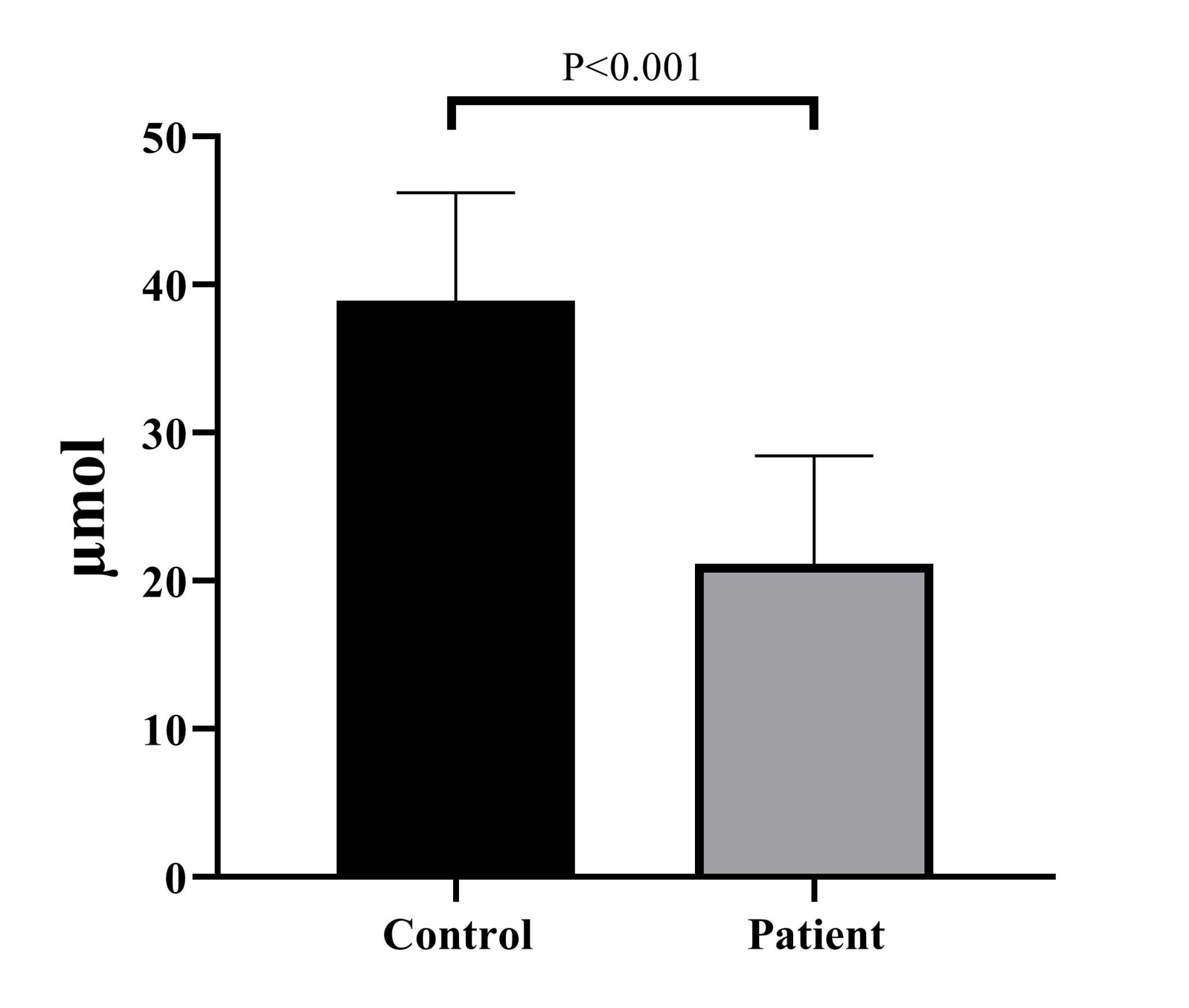
According to the results, nitrite/nitrate concentration was decreased in the patient group (21.15 ± 7.3) compared to the control group (38.91 ± 7.30). This difference was found to be statistically significant (p < 0.001) (Fig. 3).
One of the newly identified risk factors is oxidative stress, characterized by an excess of reactive oxygen species (ROS) and/or a diminished capacity for antioxidant defense. This phenomenon has been extensively documented and researched in patients with uremia.
It may play a role in the development of atherosclerosis, cardiovascular incidents, and various complications associated with CKD, including endothelial cell dysfunction, anemia, and protein-energy malnutrition [3].
Free radical reactions, particularly lipid peroxidation, are regarded as significant contributors to the development of various diseases, especially CKD [4, 5]. The progression of CKD may lead to an increase in lipid peroxidation and a reduction in antioxidant defenses, potentially resulting in oxidative damage to the kidneys of patients suffering from chronic renal failure [6]. One significant consequence of membrane lipid oxidation, primarily taking place in mitochondria, is the impairment of normal endothelial function, a critical element in cardiovascular diseases linked to CKD [7]. This phenomenon arises from the interaction of ROS with the double bonds present in unsaturated lipids, ultimately resulting in the generation and accumulation of lipid radicals. This alteration in membrane lipid structure leads to the formation of various products, including reactive electrophiles such as epoxides and aldehydes [4, 5, 8]. Malondialdehyde is a significant and primary byproduct of lipid peroxidation, serving as an important biomarker for evaluating the extent of lipid peroxidation [4, 5, 9].
Nitric oxide (NO) and its derivatives, nitrite and nitrate, serve as significant signaling molecules involved in numerous physiological and pathological conditions. Their primary functions can be categorized into three main areas:
the regulation of blood flow within the vascular endothelium (vascular homeostasis), neurotransmission, and mechanisms of host defense. The role of NO in the kidneys is multifaceted, influenced by its concentration, the specific site of release, and the duration of its action. Endothelial damage and the resulting dysfunction of the microcirculation are likely initial events in renal hypoxia, with disturbances in NO-related pathways playing crucial roles. Furthermore, NO metabolites, such as nitrite and nitrate, may serve as potential biomarkers for evaluating vascular endothelial damage in patients with CKD [10].
In recent years, research has focused on the direct correlation between changes in homocysteine metabolism and the unfavorable outcomes associated with kidney diseases. Homocysteine is a sulfur-containing non-proteinogenic amino acid that results from the demethylation of the essential amino acid methionine (Met). It serves as an intermediate in the biosynthetic pathway that facilitates the conversion of Met into cysteine. The metabolism of homocysteine takes place through
two primary pathways: remethylation and transsulfuration [11, 12, 13]. Homocysteine is recognized for its transsulfuration process, which occurs in the kidneys. Consequently, notable changes in the enzymes and mechanisms associated with transsulfuration significantly elevate plasma homocysteine levels while concurrently causing impairment in renal and cardiovascular functions. A moderate rise in plasma homocysteine levels can be detected from the early stages of CKD, coinciding with deterioration in renal function. This change is regarded as a critical element in the advancement of kidney diseases, particularly CKD [14].
Consequently, it is crucial to identify and utilize biomarkers that can facilitate the early diagnosis of kidney damage. In this research, we assessed several of these markers in individuals diagnosed with CKD.
Materials and Methods
Sample collection and preparation
The present study is a case-control study. The sample size in this study was calculated based on the significance level of 0.05 and the power of 80%, the minimum required size of 60 samples in each group. Thus, blood samples were taken from 38 male and 23 female patients diagnosed with CKD, with an average age of 58.9 ± 15.9 and 62.13 ± 13.43, respectively. At the same time, 40 healthy male individuals and 22 healthy female individuals, with an average age of 60.33 ± 10.62 and 59.3±6.64, respectively, were concurrently selected as the control group. These individuals had no prior history of any diseases, including hypertension, diabetes, infections, or kidney disease. They also had no history of smoking. Also, neither the case group nor the control group took any antioxidant drugs or other drugs that could interfere with the research parameters. All patients were 4 grade of CKD and were treated in the nephrology department of the hospital. Informed consent was obtained from the patients and subjects involved in the current study. Ethical committee approval has also been obtained. To perform tests first, 10 ml of whole blood sample was taken from each person in each group and transferred to two tubes containing EDTA-anticoagulant and without anticoagulant. Then, blood samples taken from patients were centrifuged at 4000 rpm for 10 minutes to obtain the required plasma and serum for the experiments. Hematological parameters were performed on whole blood samples.
Measurement of hematological parameters
Erythrocyte count, hematocrit, hemoglobin concentration, and other erythrocyte indices such as mean corpuscular hemoglobin (MCH), mean corpuscular hemoglobin concentration (MCHC), and mean corpuscular volume (MCV) were measured by cell counting device (Sismex, K-1000, Japan).
Measurement of biochemical parameters
All measurements of biochemical parameters, including glucose, urea, calcium, and phosphorus concentrations, were performed on a serum sample by a Chemistry autoanalyzer (Hitachi 902, Japan). Thus, the Concentration of urea was measured by the urease method, creatinine by the Jaffe reaction, calcium by the Arsenazo method, and the concentration of phosphorus was measured by the phosphomolabate method according to the kit protocol. Sodium and potassium concentrations were also measured using a flame photometer (Eppendorf, Efox 5054, Germany).
Plasma Homocysteine assessment
A plasma sample was utilized to assess homocysteine levels. The concentration of homocysteine was determined following the protocol provided by the kit (Axis-Shield, UK). This procedure involves two distinct steps.
Step 1 (Reduction reaction):
At this stage, plasma protein-bound homocysteine is first reduced to free homocysteine by tris (2-carboxyethyl) phosphine.
Step 2 (Enzymatic conversion reaction):
In this step, homocysteine is converted to cystathionine by cystathionine beta-synthase, and then cystathionine is converted to Pyruvate by cystathionine beta-lyase. Following this, pyruvate is converted to lactate by a lactate dehydrogenase enzymatic reaction (NADH is used as a cofactor in this reaction). In this reaction, the optical density (OD) of NADH to NAD converter, which is proportional to the initial concentration of homocysteine, is read using an autoanalyzer (Cobas Mira, Roche, Germany).
Malondialdehyde assessment
Malondialdehyde is the primary byproduct resulting from the peroxidation of polyunsaturated fatty acids. The concentration of malondialdehyde is measured by the reaction of thiobarbituric acid [15]. In this reaction, after the proteins were precipitated by trichloroacetic acid, the unsaturated fatty acids were oxidized with thiobarbituric acid, resulting in a color complex whose adsorption rate at 532 nm was read. This adsorption rate is converted to concentration (nmol) using a standard curve.
Measurement of nitrite/nitrate metabolites
The content of NO metabolites was assessed using the classic Griess spectro-photometric assay [16]. Briefly, the nitrate content of the sample was reduced to nitrite using vanadium trichloride. Griess reagent was added, and the samples were incubated at 37 °C for 45 minutes. Optical density (OD) was assayed using an enzyme-linked immunosorbent assay reader (Hiperion, Germany) at 540 nm. The adsorption rate is converted to concentration (µmol) using a standard curve.
Statistical Analysis
SPSS v.22.0 software (SPSS Inc., Chicago, IL, USA) was used to analyze data. The Kolmogorov-Smirnov test assayed data normal distribution. Independent sample t-test and Mann-Whitney were performed to compare groups at each time point. The results were displayed as mean standard deviation (SD), and P < 0.05 was considered significant.
Results
Hematological parameters
As indicated in Table 1, the erythrocyte count, hemoglobin levels, and hematocrit values in patients with CKD were significantly lower when compared to the control group (p < 0.01). Additionally, the erythrocyte indices, which encompass MCV, MCH, and MCHC, exhibited significant alterations in CKD patients relative to the control group (p = 0.03, p = 0.02, p = 0.01), respectively.
Biochemical parameters
The findings displayed in Table 2 indicate that the concentrations of urea, creatinine, phosphorus, and potassium were significantly elevated in individuals with CKD when compared to the control group (p = 0.0001, p = 0.0001, p = 0.001, p = 0.01), respectively. Conversely, the concentrations of sodium and calcium were significantly reduced in the CKD group relative to the control (p < 0.01).
Homocysteine concentration
The findings from the enzymatic assessment of homocysteine indicated that the concentration of homocysteine was typically elevated in patients with CKD (21.52 ± 11.69) compared to the control group (9.8 ± 4.7), which was statistically significant (p < 0.001) (Fig. 1).
Malondialdehyde concentration
The results showed an increase in malondialdehyde concentration in the CKD group (17.64 ± 6.1) compared to the control group (16.22 ± 4.66), which, according to the statistical analysis of the t-test, this difference was found to be statistically significant (p = 0.01) (Fig. 2).
NO metabolites (nitrite/nitrate)
According to the results, nitrite/nitrate concentration was decreased in the patient group (21.15 ± 7.3) compared to the control group (38.91 ± 7.30). This difference was found to be statistically significant (p < 0.001) (Fig. 3).
Table 1. Evaluation of Hematologic Parameters in CKD patients compared with the control group
| Parameters | Chronic kidney disease patients N=60 |
Control group N=60 |
P-value |
| Red blood cell (1 x106/ml) | 3.6±0.62 | 5.05±0.53 | 0.001 |
| Haemoglobin (gr/dl) | 10.65±1.92 | 14.72±1.43 | 0.001 |
| Haematocrit (%) | 32.25±5.38 | 44.90±4.04 | 0.001 |
| Mean corpuscular volume (fl) | 89.74±6.51 | 88.93±4.20 | 0.030 |
| Mean corpuscular hemoglobin (pg) | 29.59±2.65 | 29.40±2.16 | 0.020 |
| Mean corpuscular hemoglobin concentration (g/dl) | 32.79±0.89 | 32.94±1.21 | 0.010 |
| Platelet count x 1000 | 187.83±65.5 | 241.84±50 | 0.010 |
Data are presented as Mean ± SD. Differences were considered statistically significant at p < 0.05
Table 2. Evaluation of Blood Biochemical Parameters in CKD Patients Compared with the Control Group
| Parameters | Chronic kidney disease patients N=60 |
Control group N=60 |
P-value |
| Urea (mg/dl) | 141.2±52.7 | 32.4±6.7 | 0.0001 |
| Creatinine (mg/dl) | 9.6±3.2 | 1.07±0.20 | 0.0001 |
| Glucose (mg/dl) | 120.8±67.4 | 103.5±10.9 | 0.050 |
| Calcium (mg/dl) | 8.48±0.820 | 9.07±0.730 | 0.001 |
| Phosphorus (mg/dl) | 5.55±1.51 | 3.35±0.500 | 0.001 |
| Sodium (mEq/L) | 135.0±4.4 | 138.5±3.09 | 0.010 |
| Potassium (mEq/L) | 4.9±1.05 | 4.6±0.35 | 0.010 |
Data are presented as Mean ± SD. Differences were considered statistically significant at p < 0.05

Fig. 1. Comparative evaluation of homocysteine concentration between chronic kidney disease patients and control groups

Fig. 2. Comparative evaluation of malondialdehyde concentration between chronic kidney disease patients and control groups

Fig. 3. Comparative evaluation of nitric oxide metabolites concentration between chronic kidney disease patients and control groups
Discussion
CKD is a condition characterized by its widespread occurrence and significant mortality rate across various populations. One significant risk factor associated with the progression of CKD is oxidative stress, which manifests even in the initial stages of the condition. Research has demonstrated that oxidative stress exacerbates the deterioration of renal function and heightens cardiovascular risk among patients with CKD [3].
CKD is characterized by a pro-oxidant environment, leading to an excessive generation of free radicals. This overproduction adversely affects polyunsaturated fatty acids and triggers lipid peroxidation, a process that significantly contributes to the progression of various renal disorders. Malondialdehyde serves as an intermediate byproduct of lipid peroxidation and is a valuable indicator for assessing oxidative damage [9]. The findings of the present study demonstrated that the production rate of malondialdehyde was elevated in patients with CKD when compared to the healthy control group. These results agree with the high malondialdehyde levels observed by other authors in CKD patients [6]. Rasic and colleagues indicated that the concentration of malondialdehyde was elevated in patients with CKD, and their findings imply that malondialdehyde may serve as a mediator in atherosclerotic events associated with CKD [17]. Also, Rusu et al., in another study, suggests that malondialdehyde has a strong predictive value on survival in CKD patients, so regular assessment of malondialdehyde production might be useful for the evaluation of oxidative damage-related kidney injury in CKD patients [18]. The increase of lipid peroxidation in patients with CKD indicates the intensity of oxidative stress in these patients, which can be directly associated with the advancement of the disease and impairment of renal function [19].
The result of our study showed that the concentration of plasma homocysteine was increased in CKD patients compared with healthy subjects. Homocysteine could be an important biomarker of renal disease progression, mainly in CKD patients. A moderate increase in homocysteine levels can be seen in the early stage of CKD, but this increase occurs simultaneously with the decline of renal function [14]. Apparently, the high levels of plasma homocysteine found in these patients are directly related to a higher incidence of cardiovascular events.
The findings of the current study indicate an increase in homocysteine and malondialdehyde simultaneously in patients with CKD. Bayes and his colleagues, in a study, noted that an increase in plasma homocysteine increases autoxidation and the production of superoxide radicals, which in turn increases lipid peroxidation and endothelial cell toxicity, thereby increasing the risk of cardiovascular in CKD patients [20]. Increased morbidity and mortality from cardiovascular events in CKD are related to increased homocysteine, and it is among the most prevalent causes [21]. Vitamin B12 and folate play a crucial role in the proper remethylation of homocysteine. Consequently, deficiencies in these vitamins are significant contributors to hyperhomocysteinemia. Patients with CKD may experience a deficiency of these vitamins due to the catabolic state induced by renal disease, which is characterized by malnutrition, inflammation, and atherosclerosis. This condition often leads to decreased dietary intake and impaired absorption, particularly in individuals with end-stage renal disease [21]. Martella and colleagues agreed that homocysteine may serve as an indicator of renal disease progression and normal plasma level of homocysteine to ensure a better prognosis in kidney disease [14]. However, the mechanism of hyperhomocysteinemia in CKD patients is not fully understood.
NO metabolites (nitrite/nitrate) were other parameters that we measured in this study. NO is a highly reactive gaseous signaling molecule and free radical that is implicated in a variety of physiological and biochemical processes. The primary functions of the body encompass the regulation of blood circulation within the vascular endothelium (vascular homeostasis), neurotransmission, and mechanisms of host defense. Numerous experimental and clinical studies have indicated that oxidative stress and a deficiency of NO significantly play a role in the onset and advancement of renal and cardiovascular diseases [22].
In general, any condition that increases the production of free radicals, such as kidney disease, can cause a decrease in NO. The assessment of NO metabolites, specifically nitrite and nitrate, serves as an excellent indicator for evaluating the status of NO. These metabolites can be converted back into NO and other bioactive nitrogen oxides within blood and tissues. Consequently, NO and its metabolites, namely nitrite and nitrate, may serve as valuable diagnostic and prognostic biomarkers for individuals suffering from kidney disease, particularly in cases where endothelial injury and subsequent microcirculatory dysfunction are evident [10]. There is limited knowledge about NO metabolites in CKD patients. Our hypothesis in this research was that the levels of nitrite and nitrate metabolites are significantly lower in patients with CKD when compared to healthy individuals. The results confirmed this hypothesis and showed that nitrite/nitrate metabolites in CKD patients were significantly decreased compared to healthy persons.
In blood vessels, endothelial NO synthase (eNOS) represents the predominant isoform of NO synthase (NOS), and the NO produced within the endothelium serves as a crucial protective agent for the vascular system. One hypothesis suggests that in pathological conditions, such as CKD, eNOS may generate ROS, a phenomenon referred to as ‘eNOS uncoupling.’ The superoxide anion (O2) is subsequently quickly reacted with NO to produce peroxynitrite (ONOO−), which induces nitrosative stress, a significant contributor to oxidative stress [23]. Thus, impairment of endothelium-derived NO activity in oxidative stress damage will occur, resulting in an early mechanism in the pathogenesis of atherosclerosis in patients with CKD [3].
The results of this study support the notion that oxidative stress increases as stages of CKD progress, and the use of biochemical markers such as plasma homocysteine, NO metabolites (nitrite/nitrate), and malondialdehyde measurements can be helpful in identifying the severity of oxidative damage in these patients. On the other hand, by estimating the severity of oxidative damage, a more effective treatment can be determined for these patients. Considering the increase of oxidative damage in these patients, the use of antioxidant diets and vitamins may be able to reduce the oxidative damage that leads to disease progression and be included in treatment protocols.
Conclusion
The results of this study show that CKD patients are susceptible to oxidative damage that can cause disease progression and its complications, such as cardiovascular disease in these patients. Therefore, identifying markers that can indicate oxidative damage in these patients can be of particular importance in their treatment process. Evaluation of these biochemical markers in CKD helps to monitor oxidant-antioxidant status and to devise effective therapeutic strategies to reduce oxidative stress in these individuals. In this regard, antioxidant therapy may help to reduce oxidative damage and consequently increase survival in these patients. It is essential for the advancement of antioxidant therapy research in CKD to correlate measures of oxidative stress with the pathophysiological outcomes associated with the condition. Therefore, there is a need for further studies on the mechanisms of oxidative damage and the effect of antioxidants in this process.
Ethical Considerations
The patients provided their informed consent. The study was approved by the Ethics Committee of Islamic Azad University, Shiraz, Iran.
Funding
This study was supported financially by the Islamic Azad University, Shiraz, Iran.
Conflict of Interest
The authors had no conflict of interest to declare.
Acknowledgment
This project is part of the master’s thesis that was approved by the Research Council of the Islamic Azad University in 2017. We would like to thank the Iranian Blood Transfusion Research Center, a high institute for research and education in transfusion medicine. We would like to thank the Colleagues of the Biochemistry and Hematology laboratory at the Blood Transfusion Research Center, and we would like to thank the Nephrology Research Center, Tehran University of Medical Sciences, who had the necessary cooperation in this project.
Authors’ Contributions
Z.A and M.R.D participated in the study idea and design. M.R.D was the supervisor of the project, and M.R.D, M.L.P and M.T.N were the advisers for the research performance. M.R.D, Z.A, and F.K also accomplished analysis and interpretation of the results. Z.A also contributed to the literature search. All authors revised and approved the final manuscript version.
CKD is characterized by a pro-oxidant environment, leading to an excessive generation of free radicals. This overproduction adversely affects polyunsaturated fatty acids and triggers lipid peroxidation, a process that significantly contributes to the progression of various renal disorders. Malondialdehyde serves as an intermediate byproduct of lipid peroxidation and is a valuable indicator for assessing oxidative damage [9]. The findings of the present study demonstrated that the production rate of malondialdehyde was elevated in patients with CKD when compared to the healthy control group. These results agree with the high malondialdehyde levels observed by other authors in CKD patients [6]. Rasic and colleagues indicated that the concentration of malondialdehyde was elevated in patients with CKD, and their findings imply that malondialdehyde may serve as a mediator in atherosclerotic events associated with CKD [17]. Also, Rusu et al., in another study, suggests that malondialdehyde has a strong predictive value on survival in CKD patients, so regular assessment of malondialdehyde production might be useful for the evaluation of oxidative damage-related kidney injury in CKD patients [18]. The increase of lipid peroxidation in patients with CKD indicates the intensity of oxidative stress in these patients, which can be directly associated with the advancement of the disease and impairment of renal function [19].
The result of our study showed that the concentration of plasma homocysteine was increased in CKD patients compared with healthy subjects. Homocysteine could be an important biomarker of renal disease progression, mainly in CKD patients. A moderate increase in homocysteine levels can be seen in the early stage of CKD, but this increase occurs simultaneously with the decline of renal function [14]. Apparently, the high levels of plasma homocysteine found in these patients are directly related to a higher incidence of cardiovascular events.
The findings of the current study indicate an increase in homocysteine and malondialdehyde simultaneously in patients with CKD. Bayes and his colleagues, in a study, noted that an increase in plasma homocysteine increases autoxidation and the production of superoxide radicals, which in turn increases lipid peroxidation and endothelial cell toxicity, thereby increasing the risk of cardiovascular in CKD patients [20]. Increased morbidity and mortality from cardiovascular events in CKD are related to increased homocysteine, and it is among the most prevalent causes [21]. Vitamin B12 and folate play a crucial role in the proper remethylation of homocysteine. Consequently, deficiencies in these vitamins are significant contributors to hyperhomocysteinemia. Patients with CKD may experience a deficiency of these vitamins due to the catabolic state induced by renal disease, which is characterized by malnutrition, inflammation, and atherosclerosis. This condition often leads to decreased dietary intake and impaired absorption, particularly in individuals with end-stage renal disease [21]. Martella and colleagues agreed that homocysteine may serve as an indicator of renal disease progression and normal plasma level of homocysteine to ensure a better prognosis in kidney disease [14]. However, the mechanism of hyperhomocysteinemia in CKD patients is not fully understood.
NO metabolites (nitrite/nitrate) were other parameters that we measured in this study. NO is a highly reactive gaseous signaling molecule and free radical that is implicated in a variety of physiological and biochemical processes. The primary functions of the body encompass the regulation of blood circulation within the vascular endothelium (vascular homeostasis), neurotransmission, and mechanisms of host defense. Numerous experimental and clinical studies have indicated that oxidative stress and a deficiency of NO significantly play a role in the onset and advancement of renal and cardiovascular diseases [22].
In general, any condition that increases the production of free radicals, such as kidney disease, can cause a decrease in NO. The assessment of NO metabolites, specifically nitrite and nitrate, serves as an excellent indicator for evaluating the status of NO. These metabolites can be converted back into NO and other bioactive nitrogen oxides within blood and tissues. Consequently, NO and its metabolites, namely nitrite and nitrate, may serve as valuable diagnostic and prognostic biomarkers for individuals suffering from kidney disease, particularly in cases where endothelial injury and subsequent microcirculatory dysfunction are evident [10]. There is limited knowledge about NO metabolites in CKD patients. Our hypothesis in this research was that the levels of nitrite and nitrate metabolites are significantly lower in patients with CKD when compared to healthy individuals. The results confirmed this hypothesis and showed that nitrite/nitrate metabolites in CKD patients were significantly decreased compared to healthy persons.
In blood vessels, endothelial NO synthase (eNOS) represents the predominant isoform of NO synthase (NOS), and the NO produced within the endothelium serves as a crucial protective agent for the vascular system. One hypothesis suggests that in pathological conditions, such as CKD, eNOS may generate ROS, a phenomenon referred to as ‘eNOS uncoupling.’ The superoxide anion (O2) is subsequently quickly reacted with NO to produce peroxynitrite (ONOO−), which induces nitrosative stress, a significant contributor to oxidative stress [23]. Thus, impairment of endothelium-derived NO activity in oxidative stress damage will occur, resulting in an early mechanism in the pathogenesis of atherosclerosis in patients with CKD [3].
The results of this study support the notion that oxidative stress increases as stages of CKD progress, and the use of biochemical markers such as plasma homocysteine, NO metabolites (nitrite/nitrate), and malondialdehyde measurements can be helpful in identifying the severity of oxidative damage in these patients. On the other hand, by estimating the severity of oxidative damage, a more effective treatment can be determined for these patients. Considering the increase of oxidative damage in these patients, the use of antioxidant diets and vitamins may be able to reduce the oxidative damage that leads to disease progression and be included in treatment protocols.
Conclusion
The results of this study show that CKD patients are susceptible to oxidative damage that can cause disease progression and its complications, such as cardiovascular disease in these patients. Therefore, identifying markers that can indicate oxidative damage in these patients can be of particular importance in their treatment process. Evaluation of these biochemical markers in CKD helps to monitor oxidant-antioxidant status and to devise effective therapeutic strategies to reduce oxidative stress in these individuals. In this regard, antioxidant therapy may help to reduce oxidative damage and consequently increase survival in these patients. It is essential for the advancement of antioxidant therapy research in CKD to correlate measures of oxidative stress with the pathophysiological outcomes associated with the condition. Therefore, there is a need for further studies on the mechanisms of oxidative damage and the effect of antioxidants in this process.
Ethical Considerations
The patients provided their informed consent. The study was approved by the Ethics Committee of Islamic Azad University, Shiraz, Iran.
Funding
This study was supported financially by the Islamic Azad University, Shiraz, Iran.
Conflict of Interest
The authors had no conflict of interest to declare.
Acknowledgment
This project is part of the master’s thesis that was approved by the Research Council of the Islamic Azad University in 2017. We would like to thank the Iranian Blood Transfusion Research Center, a high institute for research and education in transfusion medicine. We would like to thank the Colleagues of the Biochemistry and Hematology laboratory at the Blood Transfusion Research Center, and we would like to thank the Nephrology Research Center, Tehran University of Medical Sciences, who had the necessary cooperation in this project.
Authors’ Contributions
Z.A and M.R.D participated in the study idea and design. M.R.D was the supervisor of the project, and M.R.D, M.L.P and M.T.N were the advisers for the research performance. M.R.D, Z.A, and F.K also accomplished analysis and interpretation of the results. Z.A also contributed to the literature search. All authors revised and approved the final manuscript version.
References
- Ferenbach D.A, Bonventre J.V. Acute kidney injury and chronic kidney disease: From the laboratory to the clinic. Nephrol Ther. 2016; 12 (Suppl 1): 41-8.
- Ene-Iordache B, Perico N, Bikbov B, Carminati S, Remuzzi A, Perna A, et al. Chronic kidney disease and cardiovascular risk in six regions of the world (ISN-KDDC): a cross-sectional study. Lancet Glob Health. 2016; 4(5): 307-19.
- Ling XC, 1 and Ko-Lin Kuo KL. Oxidative stress in chronic kidney disease. Renal Replacement Therapy 2018; 4(53): 1-9.
- Ayala A, Muñoz MF, Argüelles S. Lipid peroxidation: production, metabolism, and signaling mechanisms of malondialdehyde and 4-hydroxy-2-nonenal. Oxidative Medicine and Cellular Longevity 2014; 2014(1): 360438.
- Repetto M, Semprine J, Boveris A. Lipid peroxidation: chemical mechanism, biological implications, and analytical determination. Lipid peroxidation 2012; 1: 3-30.
- Rajul DK, Lalitha1 DL, and Kiranmayi P. A Study of Lipid Profile and Lipid Peroxidation in Chronic Kidney Disease with Special Reference to Hemodialysis. J Clinic Res Bioeth. 2013; 4(1): 1-5.
- Popolo A, Autore G, Pinto A, Marzocco S. Oxidative stress in patients with cardiovascular disease and chronic renal failure. Free Radical Research 2013; 47(5): 346-56.
- Fritz KS, Petersen D.R. An overview of the chemistry and biology of reactive aldehydes. Free Radical Biol and Medicine 2013; 59: 85-91.
- Grotto D, Maria LS, Valentini J, Paniz C, Schmitt G, Garcia SC, et al. Importance of the lipid peroxidation biomarkers and methodological aspects for malondialdehyde quantification. Quim. Nova 2009; 32(1):169-74.
- Mian A, Aranke M, Bryan N.S. Nitric Oxide and its Metabolites in the Critical Phase of Illness: Rapid Biomarkers in the Making. The Open Biochemistry Journal 2013; 7(1): 24-32.
- Jakubowski H. Homocysteine Modification in Protein Structure/Function and Human Disease. Physiol Rev. 2019; 99(1): 555-604.
- Jakubowski H. Homocysteine in protein structure/function and human disease: Chemical biology of homocysteine-containing proteins: Springer, 2013.
- Deyhim M.R, Khoshnaghsh F. Plasma homocysteine level and risk of thrombosis. J Appl Environ Biol Sci. 2016; 6(2): 189-93
- Martella BM, Veiga1 GRL, Alves BCA, Azzalis LA, Nqueira BC, Gehrkei FS, et al. The importance of homocysteine levels in the prognosis of patients with chronic renal disease and in hemodialysis patients. J Bras Patol Med Lab. 2018; 54(3): 170-76.
- Abuja PM, Albertini R. Methods for monitoring oxidative stress, lipid peroxidation and oxidation resistance of lipoproteins.Clin Chim Acta. 2001; 306(1-2): 1-17.
- Bryan NS, Grisham MB. Methods to detect nitric oxide and its metabolites in biological samples. Free Radical Biol Med. 2007; 43(5): 645-57.
- Rašic S, Rebic D, Hasi S Raši I, Šarac M. Influence of Malondialdehyde and Matrix Metalloproteinase-9 on Progression of Carotid Atherosclerosis in Chronic Renal Disease with Cardiometabolic Syndrome. Mediators of Inflammation 2015; 2015(1): 614357.
- Rusu CC, Racasan S, Kacso IM, Moldovan D, Potra A, Patiu IM. Malondialdehyde can predict survival in hemodialysis patients. Clujul Medical. 2016; 89(2): 250-56.
- Sridhar AVS, Rao PVNS, Sivakumar V, Satish P, Shalini P, Suchitra M, et al. Study of oxidant and antioxidant status in patients with chronic kidney disease. Journal of Clinical and Scientific Research 2018; 7(3): 124-30.
- Bayes B, Pastor MC, Bonal J, Junca J, Romero R. Homocysteine and lipid peroxidation in hemodialysis: role of folinic acid and vitamin E. Nephrol Dial Transplant 2001; 16(11): 2172-175.
- Olokor AB, Ojogwu IL, Ugbodaga PF. Hyperhomocysteinemia in Chronic Kidney Disease Patients in a Teaching Hospital in Nigeria. British Journal of Medicine and Medical Research 2016; 18(9): 1-7
- M. Carlstrom & M. F. Montenegro. Therapeutic value of stimulating the nitrate–nitrite–nitric oxide pathway to attenuate oxidative stress and restore nitric oxide bioavailability in cardiorenal disease. Journal of Internal Medicine 2019; 285: 2-10.
- Daenen K, Andries A, Mekahli D, Schepdae A.V, Jouret F, Bammens B. Oxidative stress in chronic kidney disease. Pediatric Nephrology 2019; 34: 975-91.
Type of Study: Research |
Subject:
Biochemistry
Received: 2024/07/3 | Accepted: 2024/11/12 | Published: 2025/01/13
Received: 2024/07/3 | Accepted: 2024/11/12 | Published: 2025/01/13
Send email to the article author
| Rights and permissions | |
 |
This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License. |