Mon, Apr 27, 2026
[Archive]
Volume 11, Issue 2 (May 2024)
IJML 2024, 11(2): 162-175 |
Back to browse issues page
Download citation:
BibTeX | RIS | EndNote | Medlars | ProCite | Reference Manager | RefWorks
Send citation to:



BibTeX | RIS | EndNote | Medlars | ProCite | Reference Manager | RefWorks
Send citation to:
Nejati V, Hajiesmailpoor A, Norbakhsh R, Zarabadipour M, Boroughani Z, Dalvand S, et al . Combined Impact of Wheat Germ Oil and Music Therapy on Testicular Damage Caused by Acute and Chronic Immobility Stress in Male Rats. IJML 2024; 11 (2) :162-175
URL: http://ijml.ssu.ac.ir/article-1-536-en.html
URL: http://ijml.ssu.ac.ir/article-1-536-en.html
Vahid Nejati 
 , Ayshe Hajiesmailpoor
, Ayshe Hajiesmailpoor 
 , Rahil Norbakhsh
, Rahil Norbakhsh 
 , Mahsa Zarabadipour
, Mahsa Zarabadipour 
 , Zahra Boroughani
, Zahra Boroughani 
 , Sina Dalvand
, Sina Dalvand 
 , Amin Namdari
, Amin Namdari 
 , Mehdi Shafiee Mehr
, Mehdi Shafiee Mehr 
 , Zahra Rabieefar *
, Zahra Rabieefar * 


 , Ayshe Hajiesmailpoor
, Ayshe Hajiesmailpoor 
 , Rahil Norbakhsh
, Rahil Norbakhsh 
 , Mahsa Zarabadipour
, Mahsa Zarabadipour 
 , Zahra Boroughani
, Zahra Boroughani 
 , Sina Dalvand
, Sina Dalvand 
 , Amin Namdari
, Amin Namdari 
 , Mehdi Shafiee Mehr
, Mehdi Shafiee Mehr 
 , Zahra Rabieefar *
, Zahra Rabieefar * 

Department of Histology and Embryology, Faculty of Science, Urmia University, Urmia, Iran.
Full-Text [PDF 946 kb]
(400 Downloads)
| Abstract (HTML) (661 Views)
Table 3. Effects of music and wheat germ oil on histomorphometric indexes in the study groups
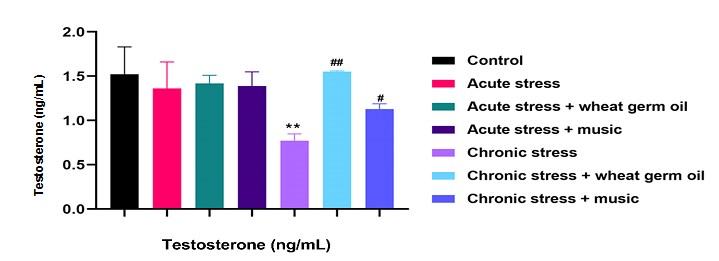
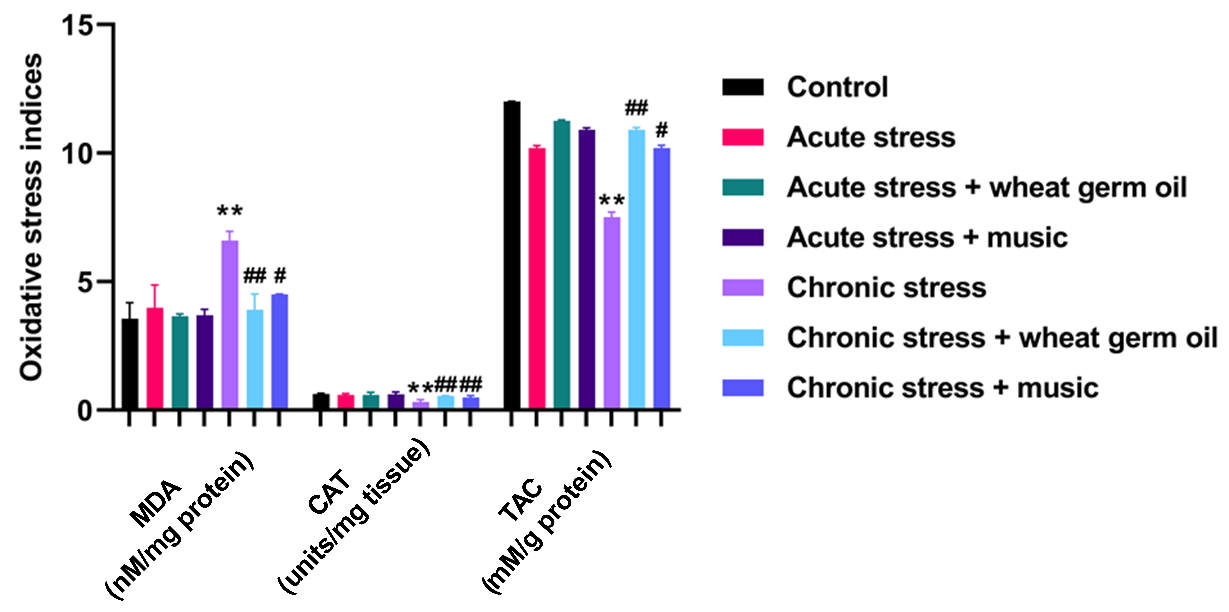
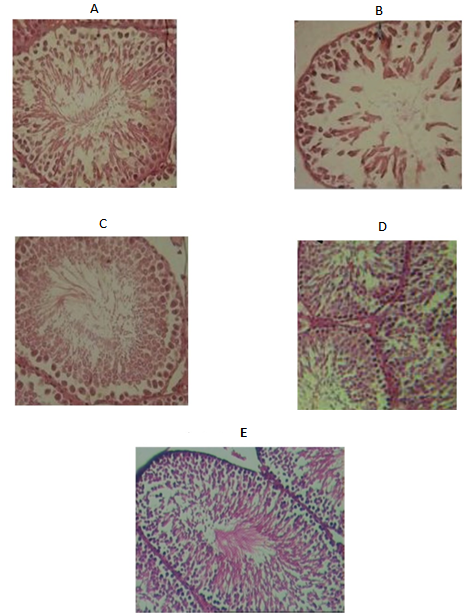
Fig. 3. Histological sections of seminiferous tubules in the study groups (H&E, X400 magnification). A: Cross-section of seminiferous tubules of the control group; B: Cross-section of seminiferous tubules of chronic stress group; C: Cross-section of seminiferous tubules of chronic stress + wheat germ oil; D: Cross-section of seminiferous tubules of chronic stress + music; E: Cross-section of seminiferous tubules of acute stress.
caused by chronic immobilization stress, we examined its effects on spermatogenesis and histomorphometric indices. Music therapy during the chronic stress period significantly normalized these parameters. Additionally, the use of nutritional therapies to manage stress has a long history. Several nutritional approaches have also been investigated in the context of male infertility [37, 38]. Wheat germ oil is a natural supplement and contains various bioactive constituents like tocopherols and tocotrienols [39]. Some experimental studies suggested that this natural supplement may act as a fertility agent [38, 40]. Similar to music therapy, wheat germ oil supplementation also significantly normalized testicular damage during the chronic stress period.
The seminiferous tubules are covered by a special and complex epithelium called the germinal epithelium or seminiferous epithelium [41, 42]. This special epithelium consists
of two groups of cells: 1) A population of non-dividing supporting cells named Sertoli cells. 2) A dividing population of germ cells that constantly migrate to the surface of the seminiferous tubules. These cells include spermatogonia, primary spermatocytes, secondary spermatocytes, and immature and mature spermatids [34, 43]. The seminiferous tubules are interconnected by interstitial tissue, which comprises loose connective tissue, lymphatic vessels, capillaries, arteries, veins, and Leydig cells [44].
The process of spermatogenesis, including the proliferation, differentiation, and maturation of germ cells, is dependent on testosterone. The health of germ cells and their ability to undergo mitotic division rely on testosterone secretion by Leydig cells. Numerous studies on various stressors have shown that stress negatively impacts sex hormone levels. For example, a 2019 study by Zou et al. demonstrated that rats exposed to chronic stress exhibited a decline in serum testosterone levels and sperm count [45]. In contrast, some studies showed the beneficial effects of music therapy on improving testosterone levels. For example, Fukui et al. found that the blood testosterone levels were significantly increased by music therapy [46]. The findings of the present study were consistent with the findings of these studies. In the present study, testosterone levels were decreased in rats exposed to chronic immobility stress, while an increase was observed in the treated group with music or wheat germ oil. Following stress, a significant decrease in testosterone levels could be one of the causes of testicular tissue changes [44, 47, 48]. In our study, rats exposed to chronic stress exhibited a decrease in the thickness of seminiferous tubules and interstitial tissue. However, these effects were significantly normalized when rats in the chronic immobility stress group were treated with wheat germ oil or exposed to music.
Reactive oxygen species (ROS) production in healthy, active sperm cells is recognized as a physiological process essential for triggering the acrosome reaction in sperm. This beneficial effect of ROS is dependent on the presence of antioxidants. Normally, antioxidant systems are present in reproductive tissues, where they prevent oxidative damage in adult gonadal cells and spermatozoa [49-51]. Excess free radicals cause cellular damage by inducing lipid peroxidation in the cell membrane and promoting oxidative damage to proteins and DNA. Mammalian sperm cells are rich in unsaturated fatty acids, plasmalogen, and sphingomyelin, which are important substrates for oxidation [52]. Many evidence revealed that an increase in free radicals has adverse effects on sperm activity and motility [4, 53]. The lipid peroxidation in the sperm membrane components decreases Na+/K+/ ATPase pump activity and ultimately a sharp decrease in sperm cell motility [54]. Recent studies have shown that infertile men's sperm plasma has lower anti-oxidant components than fertile men [55, 56]. It has been demonstrated that vitamin E, or α-tocopherol, can protect sperm parameters from the destructive effects of ROS's. Similarly, a water-soluble analog of vitamin E, Trolox, has been reported to enhance sperm motility and maintain the integrity of the mitochondrial membrane [57]. Most plants rich in anti-oxidant compounds increase sperm count, motility, and morphological sperms [58, 59]. In the present study, the mean tissue concentration of MDA indicated that chronic immobility stress significantly increased MDA levels. However, a significant decrease in MDA concentration was observed in the group treated with wheat germ oil. This reduction is likely due to the antioxidant properties of wheat germ oil [60]. Our results also showed a significant reduction in the catalase activity and total antioxidant capacity of testis tissue in the chronic stress group. These reductions were restored in the groups treated with wheat germ oil or exposed to music therapy. However, this study has some limitations. While it demonstrated the effects of music therapy and wheat germ oil on spermatogenesis indices, sperm parameters, and the oxidant/antioxidant balance in rats exposed to acute and chronic stress, it did not evaluate the impacts of these interventions on the signaling pathways involved in spermatogenesis impairment.
Conclusion
The present study results have shown that consumption of wheat germ oil or exposure to music could improve antioxidant status and enhance spermatogenesis. It seems that the increase in the number of sperm in the rats receiving wheat germ oil is most likely related to its anti-oxidant effect because anti-oxidants directly or indirectly increase the number of sperm and fertility by affecting the HPG axis. It is suggested that this study be reviewed in humans in future studies considering the beneficial effects of wheat germ oil and music on the reproductive system in male rats.
Ethics Considerations
The present investigation results from a research project of the Urmia University (UR1394/S13).
Funding
The present research was not supported by any funding organization of the commercial, nonprofit, or public sectors.
Conflict of Interests
The authors declare that there is no conflict of interest associated with this work.
Acknowledgments
The Faculty of Science of Urmia University would be greatly acknowledged for their support.
Authors’ Contribution
Vahid Nejati, Zahra Rabieefar: Participation in the implementation of methods. Ayshe Hajiesmailpoor, Amin Namdari: Participation in data analysis. Zahra Rabieefar, Rahil Norbakhsh, Sina Dalvand: Contribution in the writing of the manuscript. Vahid Nejati, Zahra Boroughani: Edit of the manuscript. Mahsa Zarabadipour: Photograph Processing. Zahra Rabieefar: Responsible for overall supervision of authors. Mehdi Shafiee Mehr: Administrative support. All authors performed editing and approving the final version of this paper for submission, participated.
References
Full-Text: (605 Views)
Introduction
Male infertility is one of the serious problems of modern human societies, especially those living in industrial societies [1]. Studies reported that the number of sperms in today's men is less than the number of them in men who lived 50 years ago [1, 2]. Problems in production, maturity, motility, and fertility of sperms are major causes of male infertility [3]. Stress exposure is one of the major causes of infertility [4]. Classically, stress is defined as a body's non-specific reaction to any nature's destructive forces, infections, and various abnormalities [5-9]. Acute stress can enhance a person's performance and be beneficial in certain situations. However, when it is prolonged or repeated, it can become harmful. Acute stress typically lasts between 5 and 30 minutes. Most people experience this type of stress at least once a month, though some individuals may experience it more frequently, depending on their job and personal circumstances [10-12]. Acute stress has a dual role; on the one hand, it causes the secretion of stress hormones such as adrenaline in the body, and on the other hand, it increases a person's tolerance in similar situations [13]. Chronic stress is not severe and can last for days, weeks, months, or even years. A variety of physiological and psychological stresses affect the hypothalamic-pituitary-adrenal (HPA) axis, the hypothalamic-pituitary-gonadal (HPG) axis, and the sympathetic system, leading to changes in some organs of the body [14, 15]. Previous studies have also shown that following acute and chronic stresses, sperm count, viability, and progressive motility as well as testosterone levels decrease in the rat testis [16, 17].
Stressful conditions with large amounts of free radicals cause an imbalance in the oxidant and antioxidant systems in the cells and tissues [18]. As the duration of stress increases, testosterone levels continue to decline, which can lead to testicular damage in some cases [19, 20]. The history of treating diseases with medicinal plants goes back to the distant past, and their use for treatment has coincided with the history of human life [21-23]. Unrefined wheat germ oil is one of the richest sources of vitamin E and α-tocopherol (a type of tocopherol with the highest vitamin E activity). Vitamin E and α-tocopherol have anti-oxidant properties [24, 25]. In the male reproductive system, this vitamin's anti-oxidant role in inhibiting free radicals' destructive effects on the testis and sperm has been reported. Besides, vitamin E can strengthen the anti-oxidant defense system of testicular cells and sperm [26-28].
Today, there is a growing emphasis on non-pharmacological methods for pain relief, known as behavioral methods. Music therapy is one such approach. Studies have demonstrated the therapeutic effects of music therapy on diabetic retinopathy, depression, mood disorders, and chronic pain in rats [29, 30].
However, the effectiveness of these therapies (herbs and music) in addressing testicular injuries caused by immobility stress has not yet been investigated. This study aimed to evaluate the impact of these two interventions on testicular damage resulting from acute and chronic immobility stress in male rats.
Materials and Methods
Thirty-five rats adult male Wistar rats weighing 230 ± 20 g were purchased from the Faculty of Veterinary Medicine, Azad University, Tabriz Branch. To adapt animals to their new environmental conditions, they were kept in conventional cages under standard conditions (12 hours of light and 12 hours of darkness) and a temperature of 22 ± 2 °C with free access to rodent diet and water. To eliminate environmental stress, the animals' needs such as light, sounds, temperature, relative humidity, and the presence of conspecifics were noticed. Animal grouping
After 14 days' adaptation period, rats were randomly divided into seven groups as follows:
Group 1: Animals in this group were considered a control group and were not affected by any exposure.
Group 2: Animals in this group were considered acute stress groups and subjected to immobility stress once for two hours.
Group 3: Animals in this group were subjected to immobility stress for 40 days (2 hours per day).
Group 4: Animals in this group were subjected to immobility stress for two hours and then received 1400 mg/kg/day of wheat germ oil orally.
Group 5: Animals in this group were subjected to immobility stress for 40 days (2 hours per day). Every time after daily stress, the rats received 1400 mg/kg/day of wheat germ oil orally.
Group 6: Animals in this group were subjected to immobility stress once for two hours. After performing stress for this group, Mozart's sonata K448 was played for 12 hours at a frequency of 70 decibels.
Group 7: Animals in this group were subjected to immobility stress for 40 days (2 hours per day). Every time after daily stress, Mozart's sonata k448 was played for this group for 12 hours at a frequency of 70 decibels.
Sample collection
At the end of the treatment period, all animals were sacrificed, 1 ml fasting blood samples were directly taken from the animal's heart (n = 5/group) using an insulin syringe, centrifuged (3000 rpm, 15 minutes), and serum samples were utilized to biochemical analyses. Subsequently, all animals were immediately weighed and the testes were removed from the body. For each rat, one of the testes was placed in a fixative container (10% formalin solution) for further histological study. Another testicle was homogenized (Heidolph, Germany) in phosphate-buffered saline (PBS, 100 mM, pH: 7.4), centrifuged at 15 min × 3 times and 4 °C, and finally kept at − 70 °C until further analysis.
Determining the amount of testosterone in the serum
Briefly, 10 μL serum samples of each rat (n=5/each group) were utilized to quantify the concentrations of testosterone using a commercial enzyme-linked immunosorbent assay (ELISA) kit (Mono-bind, CAT no: 3725-300A, USA, sensitivity: 0.038 ng/mL) following the manufacturer's protocol.
Catalase activity assessment
To figure out the activity of catalase, the ZellBio GmbH assay kit (CAT No. ZB-CAT-96A) was employed based on the colorimetric method. Briefly, ~100 mg testicular tissue of each rat (n=5/each group) was first homogenized with 100 mM PBS buffer (1 mL) and then centrifuged at 10000 rpm for 10 min. The supernatant was then used for evaluating Catalase activity using a spectrophotometer 240 nm. In this protocol, the catalase present in the sample induces the convention of hydrogen peroxide (H2O2) substrate to water and oxygen. The rate of H2O2 decomposition into water and oxygen is proportional to the catalase activity.
Lipid peroxidation assessment
Briefly, malondialdehyde (MDA), the end product of lipid peroxidation, is measured in the homogenate of testes of each rat (n = 5/each group) by its exposure to thiobarbituric acid (TBA). MDA reacts with TBA and produces thiobarbituric acid reactive substances (TBARS), a colored product. Then, the maximum absorption was measured by spectrophotometry at 523 nm as the procedure described by Ohkawa et al., 1979. The TBARS concentration was determined using a standard curve created with known MDA amounts (nM per mg protein).
Ferric reducing antioxidant power (FRAP) determination
The anti-oxidant capacity of testicular tissue was measured using the FRAP test by reducing Fe3+ (colorless) to Fe2+- Tripyridyltriazine compound (blue color) following the protocols described by Benzie [31, 32]. The increase in reaction absorbance was read in a microplate reader at 593 nm. FeSO4 was employed as a standard, and values were presented as mM/gr of wet testes tissue in this method.
Sperm morphology
Slides were stained with eosin-nigrosin to evaluate the morphology of sperm. For each sample, 100 sperms with × 1000 magnification under a light microscope were examined; sperm abnormalities were expressed as a percentage. Sperm motility was assessed according to World Health Organization guidelines. Briefly, ten μl of the culture medium and sperm mixture was placed on a special slide to evaluate sperm motility. Five microscopic fields were examined to evaluate the motility of at least 200 sperm from each sample. The membrane of live sperm is impermeable to different colors, while dead sperm absorb the color. To evaluate the percentage of live sperm, the eosin-nigrosin staining method was used. If the sperm is alive, the head and body will be white, while the dead sperm will be pink. By calculating the unstained sperms to the total available sperms, the percentage of live sperms is obtained.
Evaluation of spermatogenesis in testicular tissue
About 100 seminiferous tubules in each testis tissue were used to evaluate the following indicators. Tubular Differentiation Index (TDI), Spermiogenesis Index (SPI), and Meiosis Index (MI) were investigated to evaluate the spermatogenesis indexes in the seminiferous tubules. For TDI determination, the percentage of seminiferous tubules containing three or more differentiated spermatogenesis cells, including interstitial spermatogonia, spermatogonia type B, spermatocytes, and spermatids, were calculated. These indicate the vitality and differentiation of spermatogonia type A. For calculating the SPI, the ratio of seminiferous tubules containing sperm to seminiferous tubules without sperm was measured. The ratio of the number of round spermatids to primary spermatocytes was determined to calculate the MI.
Histomofometric evaluation
The epithelium's thickness of the seminiferous tubules and interstitial tissue space were calculated based on a micrometer with a calibrated lens.
Tissue histology
After removing testes from the rat's body, the tissues were fixed in paraformaldehyde, dehydrated in ethanol (70-100%), and then embedded in paraffin blocks. Then, five μm thick sections were stained with hematoxylin and eosin (H&E) and examined under light microscopy.
Statistical analysis
Statistical calculation was performed using SPSS software version 17. The values were presented as mean ± SEM and a one-way analysis of variance followed by Tukey post hoc test was used to compare the means between groups. p < 0.05 was accepted as a significant difference.
Results
Changes in body weight
Considering that body weight is an important indicator of animal health, we investigated the potential effects of immobility stress on body weight. All groups were weighed at the beginning and end of the study, and the results are presented in Table 1. The findings showed no significant change in body weight under acute immobility stress compared to the control group. Since the acute immobility stress was applied for only one day, no weight change was expected during this period. In the chronic immobility stress group, the final body weight showed a significant decrease compared to the control group. However, following intervention with music and wheat germ oil, body weight significantly increased.
Spermatogenesis indexes
The results did not show a change in TDI, SPI, and MI levels between study groups under acute stress (p ˃ 0.05). The rats with chronic stress showed a significant reduction in TDI, SPI, and MI than the control group (p < 0.01). When the rats were exposed to music or wheat germ oil for 40 days, all indexes were increased compared to the group of chronic immobility stress (p < 0.05 and p < 0.01; Table 2).
Testicular histomorphometric evaluation
The findings related to the possible effects of music therapy or wheat germ oil on histomorphometric indexes in the rats of immobility stress groups were presented in Table 3. In the chronic immobility stress group, the diameter of seminiferous tubules and thickness of the intermediate tissue was significantly declined than the control group, but the epithelium's thickness was increased compared to the control group. After intervention with music or wheat germ oil, all histomorphometric indexes were significantly normalized (p < 0.05 and p < 0.01; Table 3). However, no changes in these indices were observed between different acute stress groups (p ˃ 0.05, Table 3).
Sperm parameters evaluation
The results related to the effect of wheat germ oil and music on sperm parameters in rats exposed to acute and chronic immobility stress are presented in Table 4. These findings show that exposure to chronic immobility stress decreased sperm parameters compared to the control rats (p < 0.05). When the rats were exposed to music or wheat germ oil, these parameters were normalized compared to the chronic immobility stress group (p < 0.05; Table 4). However, treatment with wheat germ oil and music in rats in the acute stress group did not cause any change in sperm parameters between the study groups (p > 0.05; Table 4).
Stressful conditions with large amounts of free radicals cause an imbalance in the oxidant and antioxidant systems in the cells and tissues [18]. As the duration of stress increases, testosterone levels continue to decline, which can lead to testicular damage in some cases [19, 20]. The history of treating diseases with medicinal plants goes back to the distant past, and their use for treatment has coincided with the history of human life [21-23]. Unrefined wheat germ oil is one of the richest sources of vitamin E and α-tocopherol (a type of tocopherol with the highest vitamin E activity). Vitamin E and α-tocopherol have anti-oxidant properties [24, 25]. In the male reproductive system, this vitamin's anti-oxidant role in inhibiting free radicals' destructive effects on the testis and sperm has been reported. Besides, vitamin E can strengthen the anti-oxidant defense system of testicular cells and sperm [26-28].
Today, there is a growing emphasis on non-pharmacological methods for pain relief, known as behavioral methods. Music therapy is one such approach. Studies have demonstrated the therapeutic effects of music therapy on diabetic retinopathy, depression, mood disorders, and chronic pain in rats [29, 30].
However, the effectiveness of these therapies (herbs and music) in addressing testicular injuries caused by immobility stress has not yet been investigated. This study aimed to evaluate the impact of these two interventions on testicular damage resulting from acute and chronic immobility stress in male rats.
Materials and Methods
Thirty-five rats adult male Wistar rats weighing 230 ± 20 g were purchased from the Faculty of Veterinary Medicine, Azad University, Tabriz Branch. To adapt animals to their new environmental conditions, they were kept in conventional cages under standard conditions (12 hours of light and 12 hours of darkness) and a temperature of 22 ± 2 °C with free access to rodent diet and water. To eliminate environmental stress, the animals' needs such as light, sounds, temperature, relative humidity, and the presence of conspecifics were noticed. Animal grouping
After 14 days' adaptation period, rats were randomly divided into seven groups as follows:
Group 1: Animals in this group were considered a control group and were not affected by any exposure.
Group 2: Animals in this group were considered acute stress groups and subjected to immobility stress once for two hours.
Group 3: Animals in this group were subjected to immobility stress for 40 days (2 hours per day).
Group 4: Animals in this group were subjected to immobility stress for two hours and then received 1400 mg/kg/day of wheat germ oil orally.
Group 5: Animals in this group were subjected to immobility stress for 40 days (2 hours per day). Every time after daily stress, the rats received 1400 mg/kg/day of wheat germ oil orally.
Group 6: Animals in this group were subjected to immobility stress once for two hours. After performing stress for this group, Mozart's sonata K448 was played for 12 hours at a frequency of 70 decibels.
Group 7: Animals in this group were subjected to immobility stress for 40 days (2 hours per day). Every time after daily stress, Mozart's sonata k448 was played for this group for 12 hours at a frequency of 70 decibels.
Sample collection
At the end of the treatment period, all animals were sacrificed, 1 ml fasting blood samples were directly taken from the animal's heart (n = 5/group) using an insulin syringe, centrifuged (3000 rpm, 15 minutes), and serum samples were utilized to biochemical analyses. Subsequently, all animals were immediately weighed and the testes were removed from the body. For each rat, one of the testes was placed in a fixative container (10% formalin solution) for further histological study. Another testicle was homogenized (Heidolph, Germany) in phosphate-buffered saline (PBS, 100 mM, pH: 7.4), centrifuged at 15 min × 3 times and 4 °C, and finally kept at − 70 °C until further analysis.
Determining the amount of testosterone in the serum
Briefly, 10 μL serum samples of each rat (n=5/each group) were utilized to quantify the concentrations of testosterone using a commercial enzyme-linked immunosorbent assay (ELISA) kit (Mono-bind, CAT no: 3725-300A, USA, sensitivity: 0.038 ng/mL) following the manufacturer's protocol.
Catalase activity assessment
To figure out the activity of catalase, the ZellBio GmbH assay kit (CAT No. ZB-CAT-96A) was employed based on the colorimetric method. Briefly, ~100 mg testicular tissue of each rat (n=5/each group) was first homogenized with 100 mM PBS buffer (1 mL) and then centrifuged at 10000 rpm for 10 min. The supernatant was then used for evaluating Catalase activity using a spectrophotometer 240 nm. In this protocol, the catalase present in the sample induces the convention of hydrogen peroxide (H2O2) substrate to water and oxygen. The rate of H2O2 decomposition into water and oxygen is proportional to the catalase activity.
Lipid peroxidation assessment
Briefly, malondialdehyde (MDA), the end product of lipid peroxidation, is measured in the homogenate of testes of each rat (n = 5/each group) by its exposure to thiobarbituric acid (TBA). MDA reacts with TBA and produces thiobarbituric acid reactive substances (TBARS), a colored product. Then, the maximum absorption was measured by spectrophotometry at 523 nm as the procedure described by Ohkawa et al., 1979. The TBARS concentration was determined using a standard curve created with known MDA amounts (nM per mg protein).
Ferric reducing antioxidant power (FRAP) determination
The anti-oxidant capacity of testicular tissue was measured using the FRAP test by reducing Fe3+ (colorless) to Fe2+- Tripyridyltriazine compound (blue color) following the protocols described by Benzie [31, 32]. The increase in reaction absorbance was read in a microplate reader at 593 nm. FeSO4 was employed as a standard, and values were presented as mM/gr of wet testes tissue in this method.
Sperm morphology
Slides were stained with eosin-nigrosin to evaluate the morphology of sperm. For each sample, 100 sperms with × 1000 magnification under a light microscope were examined; sperm abnormalities were expressed as a percentage. Sperm motility was assessed according to World Health Organization guidelines. Briefly, ten μl of the culture medium and sperm mixture was placed on a special slide to evaluate sperm motility. Five microscopic fields were examined to evaluate the motility of at least 200 sperm from each sample. The membrane of live sperm is impermeable to different colors, while dead sperm absorb the color. To evaluate the percentage of live sperm, the eosin-nigrosin staining method was used. If the sperm is alive, the head and body will be white, while the dead sperm will be pink. By calculating the unstained sperms to the total available sperms, the percentage of live sperms is obtained.
Evaluation of spermatogenesis in testicular tissue
About 100 seminiferous tubules in each testis tissue were used to evaluate the following indicators. Tubular Differentiation Index (TDI), Spermiogenesis Index (SPI), and Meiosis Index (MI) were investigated to evaluate the spermatogenesis indexes in the seminiferous tubules. For TDI determination, the percentage of seminiferous tubules containing three or more differentiated spermatogenesis cells, including interstitial spermatogonia, spermatogonia type B, spermatocytes, and spermatids, were calculated. These indicate the vitality and differentiation of spermatogonia type A. For calculating the SPI, the ratio of seminiferous tubules containing sperm to seminiferous tubules without sperm was measured. The ratio of the number of round spermatids to primary spermatocytes was determined to calculate the MI.
Histomofometric evaluation
The epithelium's thickness of the seminiferous tubules and interstitial tissue space were calculated based on a micrometer with a calibrated lens.
Tissue histology
After removing testes from the rat's body, the tissues were fixed in paraformaldehyde, dehydrated in ethanol (70-100%), and then embedded in paraffin blocks. Then, five μm thick sections were stained with hematoxylin and eosin (H&E) and examined under light microscopy.
Statistical analysis
Statistical calculation was performed using SPSS software version 17. The values were presented as mean ± SEM and a one-way analysis of variance followed by Tukey post hoc test was used to compare the means between groups. p < 0.05 was accepted as a significant difference.
Results
Changes in body weight
Considering that body weight is an important indicator of animal health, we investigated the potential effects of immobility stress on body weight. All groups were weighed at the beginning and end of the study, and the results are presented in Table 1. The findings showed no significant change in body weight under acute immobility stress compared to the control group. Since the acute immobility stress was applied for only one day, no weight change was expected during this period. In the chronic immobility stress group, the final body weight showed a significant decrease compared to the control group. However, following intervention with music and wheat germ oil, body weight significantly increased.
Spermatogenesis indexes
The results did not show a change in TDI, SPI, and MI levels between study groups under acute stress (p ˃ 0.05). The rats with chronic stress showed a significant reduction in TDI, SPI, and MI than the control group (p < 0.01). When the rats were exposed to music or wheat germ oil for 40 days, all indexes were increased compared to the group of chronic immobility stress (p < 0.05 and p < 0.01; Table 2).
Testicular histomorphometric evaluation
The findings related to the possible effects of music therapy or wheat germ oil on histomorphometric indexes in the rats of immobility stress groups were presented in Table 3. In the chronic immobility stress group, the diameter of seminiferous tubules and thickness of the intermediate tissue was significantly declined than the control group, but the epithelium's thickness was increased compared to the control group. After intervention with music or wheat germ oil, all histomorphometric indexes were significantly normalized (p < 0.05 and p < 0.01; Table 3). However, no changes in these indices were observed between different acute stress groups (p ˃ 0.05, Table 3).
Sperm parameters evaluation
The results related to the effect of wheat germ oil and music on sperm parameters in rats exposed to acute and chronic immobility stress are presented in Table 4. These findings show that exposure to chronic immobility stress decreased sperm parameters compared to the control rats (p < 0.05). When the rats were exposed to music or wheat germ oil, these parameters were normalized compared to the chronic immobility stress group (p < 0.05; Table 4). However, treatment with wheat germ oil and music in rats in the acute stress group did not cause any change in sperm parameters between the study groups (p > 0.05; Table 4).
Table 1. Comparison of initial and final body weight of rats in study groups
| Groups | Early weight (gr) | Final weight (gr) |
| Control | 256±15.80 | 296±0.18 |
| Acute stress | 242±13.31 | 242±18.33 |
| Acute stress + wheat germ oil | 254±12.94 | 254±9.42 |
| Acute stress + music | 248 ± 11.83 | 248 ± 9.26 |
| Chronic stress | 253 ± 12.25 | 228 ± 20.61**a |
| Chronic stress + wheat germ oil | 251 ± 0.54 | 271 ± 16.40*b |
| Chronic stress + music | 254 ± 6.18 | 268 ± 32.15*b |
Values were expressed as mean ±SD. a: Significance compared to the control group, b: Significance compared to the chronic immobility stress group. * p < 0.05 and ** p < 0.01
Table 2. Effects of music and wheat germ oil on spermatogenesis indexes in the study groups
Table 2. Effects of music and wheat germ oil on spermatogenesis indexes in the study groups
| Groups | TDI (%) | SPI (%) | MI |
| Control | 94.64 ± 3.29 | 84.69 ± 5.14 | 2.06 ± 0.05 |
| Acute stress | 90.66 ± 3.48 | 83.42 ± 13.94 | 1.98 ± 0.03 |
| Acute stress + wheat germ oil | 93.26 ± 2.48 | 85.01 ± 2.18 | 2.03 ± 0.1 |
| Acute stress + music | 89.95 ± 14.56 | 79.94 ± 26.74 | 2.00 ± 0.09 |
| Chronic stress | 56.40 ± 2.44* | 47.13 ± 7.35* | 0.79 ± 0.01* |
| Chronic stress + wheat germ oil | 90.47 ± 3.55## | 79.28 ± 7.17## | 1.90 ± 0.1## |
| Chronic stress + music | 78.26 ± 4.63# | 68.92 ± 5.71# | 1.65 ± 0.03# |
All values are presented as mean±SD. *p < 0.05 compared to control group. #p < 0.05 and ##p < 0.01 compared to the chronic stress group. TDI = Tubular differentiation index; SPI = spermiogenesis index, MI= Meiotic index
Table 3. Effects of music and wheat germ oil on histomorphometric indexes in the study groups
| Groups | DST (µm) | TE (µm) | TIT (µm) |
| Control | 233.20 ± 13.74 | 43.78 ± 13.8 | 9.77 ± 2.95 |
| Acute stress | 228.61 ± 25.05 | 41.42 ± 13.94 | 7.34 ± 3.36 |
| Acute stress + wheat germ oil | 228.26 ± 24.8 | 42.01 ± 9.8 | 9.96 ± 2.69 |
| Acute stress + music | 225.95 ± 14.56 | 39.94 ± 12.74 | 8.40 ± 3.45 |
| Chronic stress | 201.15 ± 13.10* | 32.41 ± 11.51* | 20.73 ± 2.35** |
| Chronic stress + wheat germ oil | 228.51 ± 16.9# | 38.52 ± 12.6# | 13.94 ± 4.56## |
| Chronic stress + music | 227.91 ± 11.81# | 36.92 ± 10.56# | 17.31 ± 5.45# |
All data are expressed as mean ±SD. *p < 0.05 and **p < 0.05 compared to the control group. #p < 0.05 and ##p < 0.01 compared to chronic stress group. DST = Diameter of seminiferous tubules, TE = Thickness of the epithelium, TIT = Thickness of the intermediate tissue
Table 4. Effects of wheat germ oil and music on sperm parameters in the study groups
Table 4. Effects of wheat germ oil and music on sperm parameters in the study groups
| Groups | Sperm count (×106) | Motility (%) | Vitality (%) |
| Control | 203 ± 13.14 | 79 ± 4.57 | 93.62 ± 4.02 |
| Acute stress | 201 ± 15.9 | 80 ± 0.82 | 90 ± 2.68 |
| Acute stress + wheat germ oil | 205 ± 18.11 | 79 ± 2.04 | 89 ± 5.69 |
| Acute stress + music | 204 ± 11.8 | 81 ± 1.1 | 92 ± 4.36 |
| Chronic stress | 169 ± 16.88* | 42 ± 20.9 | 58 ± 1.15 |
| Chronic stress + wheat germ oil | 195 ± 10.4# | 73 ± 5.27 | 88 ± 1.25 |
| Chronic stress + music | 186 ± 6.06# | 59 ± 25.17 | 70 ± 8.1 |
All values are demonstrated as mean ±SD. *p < 0.05 compared to the control group and #p < 0.05 compared to the chronic stress group.
Biochemical parameters evaluation
The serum level of testosterone, MDA level, catalase activity, and total antioxidant capacity in the study groups are presented in Figures 1 and 2. Our results show that the chronic stress group dramatically decreased testosterone serum levels than the normal control group (p < 0.01). Testosterone level was increased in wheat germ oil and music groups than in the chronic stress group (p < 0.05; p < 0.01; Figure 1). After 40 days of treatment, rats in the chronic stress group had significantly reduced catalase activity compared to the control group (p < 0.01). Also, the FRAP test showed a reduction in total antioxidant status in testis tissues in this group (p < 0.01). However, these changes were restored after treatment with wheat germ oil and music (p < 0.05; p < 0.01; Figure 2). On the other hand, the MDA level was elevated in the chronic stress group compared to the control group (p < 0.01), which decreased after exposure to germ oil and music (p < 0.05; p < 0.01; Figure 2). There was no significant change in catalase activity, MDA level, and FRAP test between the acute stress and control groups.
Histological examination
In the control group, a normal morphologic characteristic was observed in the testes. Compared to chronic stress groups, no specific lesions were seen in the seminiferous tubules, and all spermatogenesis cell lines were organized in concentric layers (Figure 3A). In chronic stress groups, a decreased diameter of seminiferous tubules and epithelium was seen compared to the control group. All spermatogenesis cell lines had largely lost their communication, and there was no organization between these cells (Figure 3B). After treatment with wheat germ oil or music, the findings exhibited a normal histopathological feature. A cross-section of testicular tissue using H&D staining showed that wheat germ oil and music had protected testicular tissue from tissue damage. As shown in Figure 3C and Figure 3D, the spermatogenesis lineage cells completely and regularly are next to each other. Besides, in a cross-section of the seminiferous tubules of the acute immobility stress group, no specific lesions were seen in the seminiferous tubules, and all spermatogenesis cell lines were cohesively observed inside the tubes (Figure 3E).
The serum level of testosterone, MDA level, catalase activity, and total antioxidant capacity in the study groups are presented in Figures 1 and 2. Our results show that the chronic stress group dramatically decreased testosterone serum levels than the normal control group (p < 0.01). Testosterone level was increased in wheat germ oil and music groups than in the chronic stress group (p < 0.05; p < 0.01; Figure 1). After 40 days of treatment, rats in the chronic stress group had significantly reduced catalase activity compared to the control group (p < 0.01). Also, the FRAP test showed a reduction in total antioxidant status in testis tissues in this group (p < 0.01). However, these changes were restored after treatment with wheat germ oil and music (p < 0.05; p < 0.01; Figure 2). On the other hand, the MDA level was elevated in the chronic stress group compared to the control group (p < 0.01), which decreased after exposure to germ oil and music (p < 0.05; p < 0.01; Figure 2). There was no significant change in catalase activity, MDA level, and FRAP test between the acute stress and control groups.
Histological examination
In the control group, a normal morphologic characteristic was observed in the testes. Compared to chronic stress groups, no specific lesions were seen in the seminiferous tubules, and all spermatogenesis cell lines were organized in concentric layers (Figure 3A). In chronic stress groups, a decreased diameter of seminiferous tubules and epithelium was seen compared to the control group. All spermatogenesis cell lines had largely lost their communication, and there was no organization between these cells (Figure 3B). After treatment with wheat germ oil or music, the findings exhibited a normal histopathological feature. A cross-section of testicular tissue using H&D staining showed that wheat germ oil and music had protected testicular tissue from tissue damage. As shown in Figure 3C and Figure 3D, the spermatogenesis lineage cells completely and regularly are next to each other. Besides, in a cross-section of the seminiferous tubules of the acute immobility stress group, no specific lesions were seen in the seminiferous tubules, and all spermatogenesis cell lines were cohesively observed inside the tubes (Figure 3E).

Fig. 1. Effects of wheat germ oil and music on testosterone level in the study groups. All results are showed as mean ±SD. **p<0.01 compared to control group. #p<0.05 and ##p<0.01 compared to chronic stress group.

Fig. 2. Effects of wheat germ oil and music on oxidative stress indices in the study groups. All results are showed as mean ±SD. **p<0.01 compared to control group. #p<0.05 and ##p<0.01 compared to chronic stress group. CAT= Catalase; MDA= Malondialdehyde; TAC= Total anti-oxidant capacity.

Fig. 3. Histological sections of seminiferous tubules in the study groups (H&E, X400 magnification). A: Cross-section of seminiferous tubules of the control group; B: Cross-section of seminiferous tubules of chronic stress group; C: Cross-section of seminiferous tubules of chronic stress + wheat germ oil; D: Cross-section of seminiferous tubules of chronic stress + music; E: Cross-section of seminiferous tubules of acute stress.
Discussion
In the current study, our data demonstrated that chronic stress, but not acute stress, caused a significant decline in body weight, sperm parameters, spermatogenesis indices, morphometric indices, antioxidant activity, and serum testosterone levels. Additionally, chronic stress significantly increases the oxidative stress index. Conversely, rats treated with wheat germ oil or exposed to music showed restoration of these parameters. Because acute immobility stress was induced for only one day, none of the studied parameters were significantly affected during this period and remained constant.
Studying the impact of psychological stress on male reproductive health is highly valuable. Previous research reported that work- and depression-related stress were associated with reduced sperm concentration and quality in Chinese men [33, 34]. Their results were following other studies [35]. In our study, we observed impaired spermatogenesis in rats exposed to chronic stress. Chronic stress over 40 days significantly disrupted spermatogenesis, reduced histomorphometric indices, and decreased sperm count and motility. Various strategies have been proposed to manage chronic stress, with music therapy being one of the most successful. It has been effectively employed to reduce stress responses caused by adverse physiological and psychological factors in diverse clinical populations [36]. To evaluate whether music therapy can serve as an effective adjuvant in alleviating testicular injurycaused by chronic immobilization stress, we examined its effects on spermatogenesis and histomorphometric indices. Music therapy during the chronic stress period significantly normalized these parameters. Additionally, the use of nutritional therapies to manage stress has a long history. Several nutritional approaches have also been investigated in the context of male infertility [37, 38]. Wheat germ oil is a natural supplement and contains various bioactive constituents like tocopherols and tocotrienols [39]. Some experimental studies suggested that this natural supplement may act as a fertility agent [38, 40]. Similar to music therapy, wheat germ oil supplementation also significantly normalized testicular damage during the chronic stress period.
The seminiferous tubules are covered by a special and complex epithelium called the germinal epithelium or seminiferous epithelium [41, 42]. This special epithelium consists
of two groups of cells: 1) A population of non-dividing supporting cells named Sertoli cells. 2) A dividing population of germ cells that constantly migrate to the surface of the seminiferous tubules. These cells include spermatogonia, primary spermatocytes, secondary spermatocytes, and immature and mature spermatids [34, 43]. The seminiferous tubules are interconnected by interstitial tissue, which comprises loose connective tissue, lymphatic vessels, capillaries, arteries, veins, and Leydig cells [44].
The process of spermatogenesis, including the proliferation, differentiation, and maturation of germ cells, is dependent on testosterone. The health of germ cells and their ability to undergo mitotic division rely on testosterone secretion by Leydig cells. Numerous studies on various stressors have shown that stress negatively impacts sex hormone levels. For example, a 2019 study by Zou et al. demonstrated that rats exposed to chronic stress exhibited a decline in serum testosterone levels and sperm count [45]. In contrast, some studies showed the beneficial effects of music therapy on improving testosterone levels. For example, Fukui et al. found that the blood testosterone levels were significantly increased by music therapy [46]. The findings of the present study were consistent with the findings of these studies. In the present study, testosterone levels were decreased in rats exposed to chronic immobility stress, while an increase was observed in the treated group with music or wheat germ oil. Following stress, a significant decrease in testosterone levels could be one of the causes of testicular tissue changes [44, 47, 48]. In our study, rats exposed to chronic stress exhibited a decrease in the thickness of seminiferous tubules and interstitial tissue. However, these effects were significantly normalized when rats in the chronic immobility stress group were treated with wheat germ oil or exposed to music.
Reactive oxygen species (ROS) production in healthy, active sperm cells is recognized as a physiological process essential for triggering the acrosome reaction in sperm. This beneficial effect of ROS is dependent on the presence of antioxidants. Normally, antioxidant systems are present in reproductive tissues, where they prevent oxidative damage in adult gonadal cells and spermatozoa [49-51]. Excess free radicals cause cellular damage by inducing lipid peroxidation in the cell membrane and promoting oxidative damage to proteins and DNA. Mammalian sperm cells are rich in unsaturated fatty acids, plasmalogen, and sphingomyelin, which are important substrates for oxidation [52]. Many evidence revealed that an increase in free radicals has adverse effects on sperm activity and motility [4, 53]. The lipid peroxidation in the sperm membrane components decreases Na+/K+/ ATPase pump activity and ultimately a sharp decrease in sperm cell motility [54]. Recent studies have shown that infertile men's sperm plasma has lower anti-oxidant components than fertile men [55, 56]. It has been demonstrated that vitamin E, or α-tocopherol, can protect sperm parameters from the destructive effects of ROS's. Similarly, a water-soluble analog of vitamin E, Trolox, has been reported to enhance sperm motility and maintain the integrity of the mitochondrial membrane [57]. Most plants rich in anti-oxidant compounds increase sperm count, motility, and morphological sperms [58, 59]. In the present study, the mean tissue concentration of MDA indicated that chronic immobility stress significantly increased MDA levels. However, a significant decrease in MDA concentration was observed in the group treated with wheat germ oil. This reduction is likely due to the antioxidant properties of wheat germ oil [60]. Our results also showed a significant reduction in the catalase activity and total antioxidant capacity of testis tissue in the chronic stress group. These reductions were restored in the groups treated with wheat germ oil or exposed to music therapy. However, this study has some limitations. While it demonstrated the effects of music therapy and wheat germ oil on spermatogenesis indices, sperm parameters, and the oxidant/antioxidant balance in rats exposed to acute and chronic stress, it did not evaluate the impacts of these interventions on the signaling pathways involved in spermatogenesis impairment.
Conclusion
The present study results have shown that consumption of wheat germ oil or exposure to music could improve antioxidant status and enhance spermatogenesis. It seems that the increase in the number of sperm in the rats receiving wheat germ oil is most likely related to its anti-oxidant effect because anti-oxidants directly or indirectly increase the number of sperm and fertility by affecting the HPG axis. It is suggested that this study be reviewed in humans in future studies considering the beneficial effects of wheat germ oil and music on the reproductive system in male rats.
Ethics Considerations
The present investigation results from a research project of the Urmia University (UR1394/S13).
Funding
The present research was not supported by any funding organization of the commercial, nonprofit, or public sectors.
Conflict of Interests
The authors declare that there is no conflict of interest associated with this work.
Acknowledgments
The Faculty of Science of Urmia University would be greatly acknowledged for their support.
Authors’ Contribution
Vahid Nejati, Zahra Rabieefar: Participation in the implementation of methods. Ayshe Hajiesmailpoor, Amin Namdari: Participation in data analysis. Zahra Rabieefar, Rahil Norbakhsh, Sina Dalvand: Contribution in the writing of the manuscript. Vahid Nejati, Zahra Boroughani: Edit of the manuscript. Mahsa Zarabadipour: Photograph Processing. Zahra Rabieefar: Responsible for overall supervision of authors. Mehdi Shafiee Mehr: Administrative support. All authors performed editing and approving the final version of this paper for submission, participated.
References
- Choy JT, Eisenberg ML. Male infertility as a window to health. Fertility and Sterility 2018; 110(5): 810-14.
- Barratt CL, Björndahl L, De Jonge CJ, Lamb DJ, Osorio Martini F, McLachlan R, et al. The diagnosis of male infertility: an analysis of the evidence to support the development of global WHO guidance—challenges and future research opportunities. Human Reproduction Update 2017; 23(6): 660-80.
- Lotti F, Maggi M. Sexual dysfunction and male infertility. Nature Reviews Urology 2018; 15(5): 287-307.
- Bisht S, Faiq M, Tolahunase M, Dada R. Oxidative stress and male infertility. Nature Reviews Urology 2017; 14(8): 470-85.
- Hammen C, Kim EY, Eberhart NK, Brennan PA. Chronic and acute stress and the prediction of major depression in women. Depression and Anxiety 2009; 26(8): 718-23.
- Porcelli AJ, Delgado MR. Stress and decision making: effects on valuation, learning, and risk-taking. Current Opinion in Behavioral Sciences 2017; 14: 33-9.
- Musavi H, Abazari O, Barartabar Z, Kalaki-Jouybari F, Hemmati-Dinarvand M, Esmaeili P, et al. The benefits of Vitamin D in the COVID-19 pandemic: biochemical and immunological mechanisms. Archives of Physiology and Biochemistry 2020; 1(1): 1-9.
- Fattah A, Asadi A, Shayesteh MRH, Hesari FH, Jamalzehi S, Abbasi M, et al. Fertility and infertility implications in rheumatoid arthritis; state of the art. Inflammation Research 2020; 69: 721-29.
- Maleki N, Khosh Ravesh R, Salehiyeh S, Faisal Faiz A, Ebrahimi M, Sharbati A, et al. Comparative effects of estrogen and silibinin on cardiovascular risk biomarkers in ovariectomized rats. Gene 2022; 823: 146365.
- Dawson DK. Acute stress-induced (takotsubo) cardiomyopathy. Heart 2018; 104(2): 96-102.
- Van Oort J, Tendolkar I, Hermans E, Mulders P, Beckmann C, Schene A, et al. How the brain connects in response to acute stress: A review at the human brain systems level. Neuroscience & Biobehavioral Reviews 2017; 83(1): 281-97.
- Maleki N, Yavari N, Ebrahimi M, Faiz AF, Ravesh RK, Sharbati A, et al. Silibinin exerts anti-cancer activity on human ovarian cancer cells by increasing apoptosis and inhibiting epithelial-mesenchymal transition (EMT). Gene 2022; 823: 146275.
- Lenow JK, Constantino SM, Daw ND, Phelps EA. Chronic and acute stress promote overexploitation in serial decision making. Journal of Neuroscience 2017; 37(23): 5681-689.
- Mariotti A. The effects of chronic stress on health: new insights into the molecular mechanisms of brain–body communication. Future science OA. 2015; 1(3): 212-20.
- Abazari O, Shafaei Z, Divsalar A, Eslami-Moghadam M, Ghalandari B, Saboury AA, et al. Interaction of the synthesized anticancer compound of the methyl-glycine 1, 10-phenanthroline platinum nitrate with human serum albumin and human hemoglobin proteins by spectroscopy methods and molecular docking. Journal of the Iranian Chemical Society 2020; 17(7): 1601-614.
- Juárez-Rojas AL, García-Lorenzana M, Aragón-Martínez A, Gómez-Quiroz LE, del Socorro Retana-Márquez M. Intrinsic and extrinsic apoptotic pathways are involved in rat testis by cold water immersion-induced acute and chronic stress. Systems Biology in Reproductive Medicine 2015; 61(4): 211-21.
- García-Díaz EC, Gómez-Quiroz LE, Arenas-Ríos E, Aragón-Martínez A, Ibarra-Arias JA, Retana-Márquez MDSI. Oxidative status in testis and epididymal sperm parameters after acute and chronic stress by cold-water immersion in the adult rat. Systems Biology in Reproductive Medicine 2015; 61(3): 150-60.
- Sies H. On the history of oxidative stress: Concept and some aspects of current development. Current Opinion in Toxicology 2018; 7(1): 122-26.
- Asadi N, Bahmani M, Kheradmand A, Rafieian-Kopaei M. The impact of oxidative stress on testicular function and the role of antioxidants in improving it: A review. Journal of Clinical and Diagnostic Research 2017; 11(5): 1.
- Durairajanayagam D, Agarwal A, Ong C. Causes, effects and molecular mechanisms of testicular heat stress. Reproductive Biomedicine Online 2015; 30(1): 14-27.
- Jamshidi-Kia F, Lorigooini Z, Amini-Khoei H. Medicinal plants: Past history and future perspective. Journal of Herbmed Pharmacology 2018; 7(1): 1-12.
- Musavi H, Tabnak M, Sheini FA, Bezvan MH, Amidi F, Abbasi M. Effect of garlic (Allium sativum) on male fertility: a systematic review. Journal of Herbmed Pharmacology 2018; 7(4): 306-312.
- Maleki N, Ravesh RK, Salehiyeh S, Faiz AF, Ebrahimi M, Sharbati A, et al. Comparative effects of estrogen and silibinin on cardiovascular risk biomarkers in ovariectomized rats. Gene 2022; 823: 146365.
- Khadangi F, Azzi A. Vitamin E–The next 100 years. IUBMB Life 2019; 71(4): 411-15.
- Abazari O, Divsalar A, Ghobadi R. Inhibitory effects of oxali-Platin as a chemotherapeutic drug on the function and structure of bovine liver catalase. Journal of Biomolecular Structure and Dynamics 2020; 38(2): 609-15.
- Ulfanov O, Cil N, Adiguzel E. Protective effects of vitamin E on aluminium sulphate-induced testicular damage. Toxicology and Industrial Health 2020; 36(4): 215-27.
- Sabeti P, Pourmasumi S, Rahiminia T, Akyash F, Talebi AR. Etiologies of sperm oxidative stress. International Journal of Reproductive BioMedicine 2016; 14(4): 231-40.
- Alaei Sheini F, Tabnak M, Hasanzadeh Bezvan M, Mahdiannasser M, Musavi H, Choobineh H, et al. A systematic review of the evidence on the effects of cytomegalovirus on abortion. International Journal of Medical Laboratory 2018; 5(3): 173-81.
- Gao J, Chen S, Lin S, Han H. Effect of music therapy on pain behaviors in rats with bone cancer pain. J BUON. 2016; 21(2): 466-72.
- Luo C, Fan H, Li S, Zou Y. Therapeutic of candesartan and music therapy in diabetic retinopathy with depression in rats. Evidence‐Based Complementary and Alternative Medicine 2021; 2021(1): 5570356.
- Benzie IF, Strain JJ. The ferric reducing ability of plasma (FRAP) as a measure of “antioxidant power”: the FRAP assay. Analytical Biochemistry 1996; 239(1): 70-6.
- Zahra Z, Tina Nayerpour D, Armaghan L, Zakieh Sadat S, Mohammad P, Fahimeh H, et al. The effect of piperine on MMP-9, VEGF, and E-cadherin expression in breast cancer MCF-7 cell line. Basic & Clinical Cancer Research 2021; 12(3): 232-39.
- Zou P, Wang X, Sun L, Chen Q, Yang H, Zhou N, et al. Semen quality in Chinese college students: associations with depression and physical activity in a cross-sectional study. Psychosomatic Medicine 2018; 80(6): 564-72.
- Mirzaei A, Abbasi M, Sepehri S, Mirzaei M. The effects of Allium porrum and Medicago sativa on iron concentration in thalassemia serums. Life Science Journal 2013; 10(S 11): 27-31.
- Nordkap L, Jensen TK, Hansen ÅM, Lassen TH, Bang AK, Joensen UN, et al. Psychological stress and testicular function: a cross-sectional study of 1,215 Danish men. Fertility and Sterility 2016; 105(1): 174-87.
- de Witte M, Pinho ADS, Stams GJ, Moonen X, Bos AE, van Hooren S. Music therapy for stress reduction: a systematic review and meta-analysis. Health Psychology Review 2022; 16(1): 134-59.
- Shahidi M, Moradi A, Dayati P. Zingerone attenuates zearalenone-induced steroidogenesis impairment and apoptosis in TM3 Leydig cell line. Toxicon 2022; 211: 50-60.
- Hamdi H. The preventive role of wheat germ oil against sertraline‐induced testicular damage in male albino rats. Andrologia 2019; 51(10): 13369.
- Ghafoor K, Özcan MM, AL‐Juhaımı F, Babıker EE, Sarker ZI, Ahmed IAM, et al. Nutritional composition, extraction, and utilization of wheat germ oil: a review. European Journal of Lipid Science and Technology 2017; 119(7): 1600160.
- El-Sayed AI. Effect of wheat germ oil and coenzyme Q10 on physiological performance and testicular oxidative stress markers in rabbit bucks. Annals of Agricultural Science, Moshtohor 2019; 57(1): 47-58.
- Nakata H. Morphology of mouse seminiferous tubules. Anatomical Science International 2019; 94(1): 1-10.
- Pourgholi M, Abazari O, Pourgholi L, Ghasemi-Kasman M, Boroumand M. Association between rs3088440 (G> A) polymorphism at 9p21. 3 locus with the occurrence and severity of coronary artery disease in an Iranian population. Molecular Biology Reports 2021; 48(8): 5905-912.
- Sertoli E. The structure of seminiferous tubules and the development of [spermatids] in rats. Biology of Reproduction 2018; 99(3): 482.
- Zhao X, Wen X, Ji M, Guan X, Chen P, Hao X, et al. Differentiation of seminiferous tubule-associated stem cells into leydig cell and myoid cell lineages. Molecular and Cellular Endocrinology 2021: 111179.
- Zou P, Wang X, Yang W, Liu C, Chen Q, Yang H, et al. Mechanisms of stress-induced spermatogenesis impairment in male rats following unpredictable chronic mild stress (uCMS). International Journal of Molecular Sciences 2019; 20(18): 4470.
- Fukui H, Arai A, Toyoshima K. Efficacy of music therapy in treatment for the patients with Alzheimer’s disease. International Journal of Alzheimer’s Disease 2012; 2012(1): 531646.
- Chung JY, Brown S, Chen H, Liu J, Papadopoulos V, Zirkin B. Effects of pharmacologically induced Leydig cell testosterone production on intratesticular testosterone and spermatogenesis. Biology of Reproduction 2020; 102(2): 489-98.
- Panji M, Behmard V, Zare Z, Malekpour M, Nejadbiglari H, Yavari S, et al. Synergistic effects of green tea extract and paclitaxel in the induction of mitochondrial apoptosis in ovarian cancer cell lines. Gene 2021; 787: 145638.
- Agarwal A, Sengupta P. Oxidative stress and its association with male infertility. Male infertility: Springer, 2020, p. 57-68.
- Beygi Z, Forouhari S, Mahmoudi E, Hayat SM, Nourimand F. Role of oxidative stress and antioxidant supplementation in male fertility. Current Molecular Medicine 2021; 21(4): 265-82.
- Musavi H, Abazari O, Safaee MS, Variji A, Koohshekan B, Kalaki-Jouybari F, et al. Mechanisms of COVID-19 entry into the cell: potential therapeutic approaches based on virus entry inhibition in COVID-19 patients with underlying diseases. Iranian Journal of Allergy, Asthma and Immunology 2021: 1-13.
- Van Tran L, Malla BA, Kumar S, Tyagi AK. Polyunsaturated fatty acids in male ruminant reproduction: A review. Asian-Australasian Journal of Animal Sciences 2017; 30(5): 622.
- Alahmar AT. Role of oxidative stress in male infertility: An updated review. Journal of Human Reproductive Sciences 2019; 12(1): 4.
- Silva LN, Pessoa MTC, Alves SL, Venugopal J, Cortes VF, Santos HL, et al. Differences of lipid membrane modulation and oxidative stress by digoxin and 21-benzylidene digoxin. Experimental Cell Research 2017; 359(1): 291-98.
- Yazar H, Halis F, Nasir Y, Guzel D, Akdogan M, Gokce A. Effect of the oxidant-antioxidant system in seminal plasma on varicocele and idiopathic infertility in male humans. Clinical Laboratory 2017; 63(5): 935-40.
- Ambad RS, Butola MLK, Bankar N, Mahakalkar C. Oxidative stress and anti oxidant status in male infertility. European Journal of Molecular & Clinical Medicine 2021; 8(1): 340-49.
- Barik G, Chaturvedula L, Bobby Z. Role of oxidative stress and antioxidants in male infertility: An interventional study. Journal of Human Reproductive Sciences 2019; 12(3): 204.
- Zare Z, Dizaj TN, Lohrasbi A, Sheikhalishahi ZS, Asadi A, Zakeri M, et al. Silibinin inhibits TGF-β-induced MMP-2 and MMP-9 through Smad Signaling pathway in colorectal cancer HT-29 cells. Basic & Clinical Cancer Research 2020; 12(2): 79-88.
- El-Shiekh R, Al-Mahdy D, Hifnawy M, Abdel-Sattar E. In-vitro screening of selected traditional medicinal plants for their anti-obesity and anti-oxidant activities. South African Journal of Botany. 2019; 123: 43-50.
- Liaqat H, Jeong E, Kim KJ, Kim JY. Effect of wheat germ on metabolic markers: a systematic review and meta-analysis of randomized controlled trials. Food Science and Biotechnology 2020; 29(6): 739-49.
Type of Study: Research |
Subject:
Genetics/ Biotechnology
Received: 2024/12/1 | Accepted: 2025/01/5 | Published: 2025/03/9
Received: 2024/12/1 | Accepted: 2025/01/5 | Published: 2025/03/9
Send email to the article author
| Rights and permissions | |
 |
This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License. |